| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Ligand | BDBM50055123 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_4166 (CHEMBL619233) |
|---|
| IC50 | 5600±n/a nM |
|---|
| Citation |  Adams, JL; Garigipati, RS; Sorenson, M; Schmidt, SJ; Brian, WR; Newton, JF; Tyrrell, KA; Garver, E; Yodis, LA; Chabot-Fletcher, M; Tzimas, M; Webb, EF; Breton, JJ; Griswold, DE Bicyclic N-hydroxyurea inhibitors of 5-lipoxygenase: pharmacodynamic, pharmacokinetic, and in vitro metabolic studies characterizing N-hydroxy-N-(2,3-dihydro-6-(phenylmethoxy)-3-benzofuranyl)urea. J Med Chem39:5035-46 (1997) [PubMed] Article Adams, JL; Garigipati, RS; Sorenson, M; Schmidt, SJ; Brian, WR; Newton, JF; Tyrrell, KA; Garver, E; Yodis, LA; Chabot-Fletcher, M; Tzimas, M; Webb, EF; Breton, JJ; Griswold, DE Bicyclic N-hydroxyurea inhibitors of 5-lipoxygenase: pharmacodynamic, pharmacokinetic, and in vitro metabolic studies characterizing N-hydroxy-N-(2,3-dihydro-6-(phenylmethoxy)-3-benzofuranyl)urea. J Med Chem39:5035-46 (1997) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Name: | Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Synonyms: | Alox5 | Arachidonate 5-lipoxygenase | LOX5_RAT |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 78082.31 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_1432947 |
|---|
| Residue: | 673 |
|---|
| Sequence: | MPSYTVTVATGSQWFAGTDDYIYLSLIGSAGCSEKHLLDKAFYNDFERGGRDSYDVTVDE
ELGEIYLVKIEKRKYRLHDDWYLKYITLKTPHDYIEFPCYRWITGEGEIVLRDGCAKLAR
DDQIHILKQHRRKELETRQKQYRWMEWNPGFPLSIDAKCHKDLPRDIQFDSEKGVDFVLN
YSKAMENLFINRFMHMFQSSWHDFADFEKIFVKISNTISERVKNHWQEDLMFGYQFLNGC
NPVLIKRCTELPKKLPVTTEMVECSLERQLSLEQEVQEGNIFIVDYELLDGIDANKTDPC
THQFLAAPICLLYKNLANKIVPIAIQLNQTPGEKNPIFLPTDSKYDWLLAKIWVRSSDFH
IHQTITHLLRTHLVSEVFGIAMYRQLPAVHPLFKLLVAHVRFTIAINTKAREQLNCEYGL
FDKANATGGGGHVQMVQRAVQDLTYSSLCFPEAIKARGMDNTEDIPYYFYRDDGLLVWEA
IQSFTTEVVSIYYEDDQVVEEDQELQDFVKDVYVYGMRGRKASGFPKSIKSREKLSEYLT
VVIFTASAQHAAVNFGQYDWCSWIPNAPPTMRAPPPTAKGVVTIEQIVDTLPDRGRSCWH
LGAVWALSQFQENELFLGMYPEEHFIEKPVKEAMIRFRKNLEAIVSVIAERNKNKKLPYY
YLSPDRIPNSVAI
|
|
|
|---|
| BDBM50055123 |
|---|
| n/a |
|---|
| Name | BDBM50055123 |
|---|
| Synonyms: | CHEMBL358858 | N-Hydroxy-N-[2,3-dihydro-7-(phenoxy)-1H-inden-1yl]urea |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H16N2O3 |
|---|
| Mol. Mass. | 284.3098 |
|---|
| SMILES | NC(=O)N(O)C1CCc2cccc(Oc3ccccc3)c12 |
|---|
| Structure | 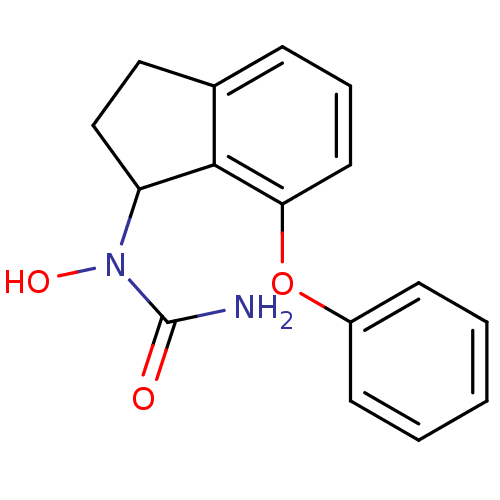
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Adams, JL; Garigipati, RS; Sorenson, M; Schmidt, SJ; Brian, WR; Newton, JF; Tyrrell, KA; Garver, E; Yodis, LA; Chabot-Fletcher, M; Tzimas, M; Webb, EF; Breton, JJ; Griswold, DE Bicyclic N-hydroxyurea inhibitors of 5-lipoxygenase: pharmacodynamic, pharmacokinetic, and in vitro metabolic studies characterizing N-hydroxy-N-(2,3-dihydro-6-(phenylmethoxy)-3-benzofuranyl)urea. J Med Chem39:5035-46 (1997) [PubMed] Article
Adams, JL; Garigipati, RS; Sorenson, M; Schmidt, SJ; Brian, WR; Newton, JF; Tyrrell, KA; Garver, E; Yodis, LA; Chabot-Fletcher, M; Tzimas, M; Webb, EF; Breton, JJ; Griswold, DE Bicyclic N-hydroxyurea inhibitors of 5-lipoxygenase: pharmacodynamic, pharmacokinetic, and in vitro metabolic studies characterizing N-hydroxy-N-(2,3-dihydro-6-(phenylmethoxy)-3-benzofuranyl)urea. J Med Chem39:5035-46 (1997) [PubMed] Article