| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Muscarinic acetylcholine receptor M2 |
|---|
| Ligand | BDBM50065211 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_139624 (CHEMBL749006) |
|---|
| IC50 | 69300.0±n/a nM |
|---|
| Citation |  Tecle, H; Barrett, SD; Lauffer, DJ; Augelli-Szafran, C; Brann, MR; Callahan, MJ; Caprathe, BW; Davis, RE; Doyle, PD; Eubanks, D; Lipiniski, W; Mirzadegan, T; Moos, WH; Moreland, DW; Nelson, CB; Pavia, MR; Raby, C; Schwarz, RD; Spencer, CJ; Thomas, AJ; Jaen, JC Design and synthesis of m1-selective muscarinic agonists: (R)-(-)-(Z)-1-Azabicyclo[2.2.1]heptan-3-one, O-(3-(3'-methoxyphenyl)-2-propynyl)oxime maleate (CI-1017), a functionally m1-selective muscarinic agonist. J Med Chem41:2524-36 (1998) [PubMed] Article Tecle, H; Barrett, SD; Lauffer, DJ; Augelli-Szafran, C; Brann, MR; Callahan, MJ; Caprathe, BW; Davis, RE; Doyle, PD; Eubanks, D; Lipiniski, W; Mirzadegan, T; Moos, WH; Moreland, DW; Nelson, CB; Pavia, MR; Raby, C; Schwarz, RD; Spencer, CJ; Thomas, AJ; Jaen, JC Design and synthesis of m1-selective muscarinic agonists: (R)-(-)-(Z)-1-Azabicyclo[2.2.1]heptan-3-one, O-(3-(3'-methoxyphenyl)-2-propynyl)oxime maleate (CI-1017), a functionally m1-selective muscarinic agonist. J Med Chem41:2524-36 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Muscarinic acetylcholine receptor M2 |
|---|
| Name: | Muscarinic acetylcholine receptor M2 |
|---|
| Synonyms: | ACM2_HUMAN | CHRM2 | Cholinergic, muscarinic M2 | Muscarinic acetylcholine receptor M2 and M4 | Muscarinic acetylcholine receptor M2 and M5 | RecName: Full=Muscarinic acetylcholine receptor M2 |
|---|
| Type: | GPCR |
|---|
| Mol. Mass.: | 51730.61 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08172 |
|---|
| Residue: | 466 |
|---|
| Sequence: | MNNSTNSSNNSLALTSPYKTFEVVFIVLVAGSLSLVTIIGNILVMVSIKVNRHLQTVNNY
FLFSLACADLIIGVFSMNLYTLYTVIGYWPLGPVVCDLWLALDYVVSNASVMNLLIISFD
RYFCVTKPLTYPVKRTTKMAGMMIAAAWVLSFILWAPAILFWQFIVGVRTVEDGECYIQF
FSNAAVTFGTAIAAFYLPVIIMTVLYWHISRASKSRIKKDKKEPVANQDPVSPSLVQGRI
VKPNNNNMPSSDDGLEHNKIQNGKAPRDPVTENCVQGEEKESSNDSTSVSAVASNMRDDE
ITQDENTVSTSLGHSKDENSKQTCIRIGTKTPKSDSCTPTNTTVEVVGSSGQNGDEKQNI
VARKIVKMTKQPAKKKPPPSREKKVTRTILAILLAFIITWAPYNVMVLINTFCAPCIPNT
VWTIGYWLCYINSTINPACYALCNATFKKTFKHLLMCHYKNIGATR
|
|
|
|---|
| BDBM50065211 |
|---|
| n/a |
|---|
| Name | BDBM50065211 |
|---|
| Synonyms: | 1-Aza-bicyclo[2.2.1]heptan-3-one O-[3-(2-methoxy-phenyl)-prop-2-ynyl]-oxime | CHEMBL86150 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H18N2O2 |
|---|
| Mol. Mass. | 270.3263 |
|---|
| SMILES | COc1ccccc1C#CCO\N=C1/CN2CCC1C2 |
|---|
| Structure | 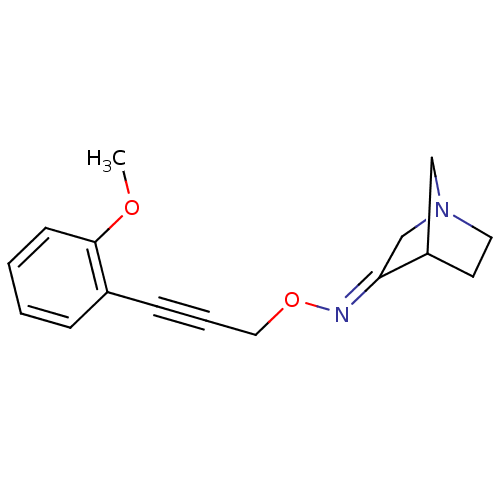
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tecle, H; Barrett, SD; Lauffer, DJ; Augelli-Szafran, C; Brann, MR; Callahan, MJ; Caprathe, BW; Davis, RE; Doyle, PD; Eubanks, D; Lipiniski, W; Mirzadegan, T; Moos, WH; Moreland, DW; Nelson, CB; Pavia, MR; Raby, C; Schwarz, RD; Spencer, CJ; Thomas, AJ; Jaen, JC Design and synthesis of m1-selective muscarinic agonists: (R)-(-)-(Z)-1-Azabicyclo[2.2.1]heptan-3-one, O-(3-(3'-methoxyphenyl)-2-propynyl)oxime maleate (CI-1017), a functionally m1-selective muscarinic agonist. J Med Chem41:2524-36 (1998) [PubMed] Article
Tecle, H; Barrett, SD; Lauffer, DJ; Augelli-Szafran, C; Brann, MR; Callahan, MJ; Caprathe, BW; Davis, RE; Doyle, PD; Eubanks, D; Lipiniski, W; Mirzadegan, T; Moos, WH; Moreland, DW; Nelson, CB; Pavia, MR; Raby, C; Schwarz, RD; Spencer, CJ; Thomas, AJ; Jaen, JC Design and synthesis of m1-selective muscarinic agonists: (R)-(-)-(Z)-1-Azabicyclo[2.2.1]heptan-3-one, O-(3-(3'-methoxyphenyl)-2-propynyl)oxime maleate (CI-1017), a functionally m1-selective muscarinic agonist. J Med Chem41:2524-36 (1998) [PubMed] Article