| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 6 |
|---|
| Ligand | BDBM50090527 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_3623 |
|---|
| Ki | 0.600±n/a nM |
|---|
| Citation |  Isaac, M; Slassi, A; Xin, T; MacLean, N; Wilson, J; McCallum, K; Wang, H; Demchyshyn, L 6-Bicyclopiperazinyl-1-arylsulfonylindoles and 6-bicyclopiperidinyl-1-arylsulfonylindoles derivatives as novel, potent, and selective 5-HT6 receptor antagonists. Bioorg Med Chem Lett10:1719-21 (2000) [PubMed] Isaac, M; Slassi, A; Xin, T; MacLean, N; Wilson, J; McCallum, K; Wang, H; Demchyshyn, L 6-Bicyclopiperazinyl-1-arylsulfonylindoles and 6-bicyclopiperidinyl-1-arylsulfonylindoles derivatives as novel, potent, and selective 5-HT6 receptor antagonists. Bioorg Med Chem Lett10:1719-21 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 6 |
|---|
| Name: | 5-hydroxytryptamine receptor 6 |
|---|
| Synonyms: | 5-HT-6 | 5-HT6 | 5-hydroxytryptamine receptor 6 (5-HT-6) | 5-hydroxytryptamine receptor 6 (5-HT6R) | 5-hydroxytryptamine receptor 6 (5HT6) | 5HT6R_HUMAN | HTR6 | Serotonin (5-HT3) receptor | Serotonin 6 (5-HT6) receptor | Serotonin Receptor 6 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46968.43 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P50406 |
|---|
| Residue: | 440 |
|---|
| Sequence: | MVPEPGPTANSTPAWGAGPPSAPGGSGWVAAALCVVIALTAAANSLLIALICTQPALRNT
SNFFLVSLFTSDLMVGLVVMPPAMLNALYGRWVLARGLCLLWTAFDVMCCSASILNLCLI
SLDRYLLILSPLRYKLRMTPLRALALVLGAWSLAALASFLPLLLGWHELGHARPPVPGQC
RLLASLPFVLVASGLTFFLPSGAICFTYCRILLAARKQAVQVASLTTGMASQASETLQVP
RTPRPGVESADSRRLATKHSRKALKASLTLGILLGMFFVTWLPFFVANIVQAVCDCISPG
LFDVLTWLGYCNSTMNPIIYPLFMRDFKRALGRFLPCPRCPRERQASLASPSLRTSHSGP
RPGLSLQQVLPLPLPPDSDSDSDAGSGGSSGLRLTAQLLLPGEATQDPPLPTRAAAAVNF
FNIDPAEPELRPHPLGIPTN
|
|
|
|---|
| BDBM50090527 |
|---|
| n/a |
|---|
| Name | BDBM50090527 |
|---|
| Synonyms: | 1-(Naphthalene-1-sulfonyl)-6-(octahydro-indolizin-7-yl)-1H-indole | CHEMBL299822 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H26N2O2S |
|---|
| Mol. Mass. | 430.562 |
|---|
| SMILES | O=S(=O)(c1cccc2ccccc12)n1ccc2ccc(cc12)C1CCN2CCCC2C1 |
|---|
| Structure | 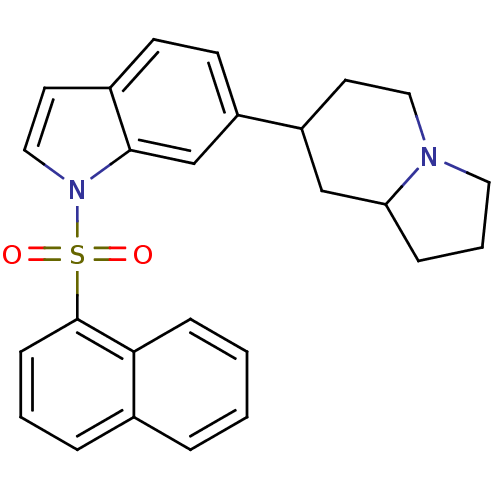
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Isaac, M; Slassi, A; Xin, T; MacLean, N; Wilson, J; McCallum, K; Wang, H; Demchyshyn, L 6-Bicyclopiperazinyl-1-arylsulfonylindoles and 6-bicyclopiperidinyl-1-arylsulfonylindoles derivatives as novel, potent, and selective 5-HT6 receptor antagonists. Bioorg Med Chem Lett10:1719-21 (2000) [PubMed]
Isaac, M; Slassi, A; Xin, T; MacLean, N; Wilson, J; McCallum, K; Wang, H; Demchyshyn, L 6-Bicyclopiperazinyl-1-arylsulfonylindoles and 6-bicyclopiperidinyl-1-arylsulfonylindoles derivatives as novel, potent, and selective 5-HT6 receptor antagonists. Bioorg Med Chem Lett10:1719-21 (2000) [PubMed]