| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Somatostatin receptor type 2 |
|---|
| Ligand | BDBM50537064 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1935684 (CHEMBL4481443) |
|---|
| Ki | 0.140±n/a nM |
|---|
| Citation |  White, BH; Whalen, K; Kriksciukaite, K; Alargova, R; Au Yeung, T; Bazinet, P; Brockman, A; DuPont, M; Oller, H; Lemelin, CA; Lim Soo, P; Moreau, B; Perino, S; Quinn, JM; Sharma, G; Shinde, R; Sweryda-Krawiec, B; Wooster, R; Bilodeau, MT Discovery of an SSTR2-Targeting Maytansinoid Conjugate (PEN-221) with Potent Activity in Vitro and in Vivo. J Med Chem62:2708-2719 (2019) [PubMed] Article White, BH; Whalen, K; Kriksciukaite, K; Alargova, R; Au Yeung, T; Bazinet, P; Brockman, A; DuPont, M; Oller, H; Lemelin, CA; Lim Soo, P; Moreau, B; Perino, S; Quinn, JM; Sharma, G; Shinde, R; Sweryda-Krawiec, B; Wooster, R; Bilodeau, MT Discovery of an SSTR2-Targeting Maytansinoid Conjugate (PEN-221) with Potent Activity in Vitro and in Vivo. J Med Chem62:2708-2719 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Somatostatin receptor type 2 |
|---|
| Name: | Somatostatin receptor type 2 |
|---|
| Synonyms: | SOMATOSTATIN SST2 | SRIF-1 | SS-2-R | SS2-R | SS2R | SSR2_HUMAN | SSTR2 | Somatostatin receptor type 2 (SSTR2) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 41344.94 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P30874 |
|---|
| Residue: | 369 |
|---|
| Sequence: | MDMADEPLNGSHTWLSIPFDLNGSVVSTNTSNQTEPYYDLTSNAVLTFIYFVVCIIGLCG
NTLVIYVILRYAKMKTITNIYILNLAIADELFMLGLPFLAMQVALVHWPFGKAICRVVMT
VDGINQFTSIFCLTVMSIDRYLAVVHPIKSAKWRRPRTAKMITMAVWGVSLLVILPIMIY
AGLRSNQWGRSSCTINWPGESGAWYTGFIIYTFILGFLVPLTIICLCYLFIIIKVKSSGI
RVGSSKRKKSEKKVTRMVSIVVAVFIFCWLPFYIFNVSSVSMAISPTPALKGMFDFVVVL
TYANSCANPILYAFLSDNFKKSFQNVLCLVKVSGTDDGERSDSKQDKSRLNETTETQRTL
LNGDLQTSI
|
|
|
|---|
| BDBM50537064 |
|---|
| n/a |
|---|
| Name | BDBM50537064 |
|---|
| Synonyms: | CHEMBL4563111 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C123H167ClN20O29S4 |
|---|
| Mol. Mass. | 2553.472 |
|---|
| SMILES | [H][C@@]12O[C@]1(C)[C@H](CC(=O)N(C)c1cc(C\C(C)=C\C=C\[C@@H](OC)[C@@]3(O)C[C@]([H])(OC(=O)N3)[C@H]2C)cc(OC)c1Cl)OC(=O)[C@H](C)N(C)C(=O)CCSC1CC(=O)N(CCCNC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc2ccccc2)NC(=O)CNC(=O)CCCCCN2C(=O)CC(SCCCN[C@H](Cc3ccccc3)C(=O)N[C@H]3CSSC[C@H](NC(=O)[C@@]([H])(NC(=O)[C@H](CCCCN)NC(=O)[C@@H](Cc4c[nH]c5ccccc45)NC(=O)[C@H](Cc4ccccc4)NC3=O)[C@@H](C)O)C(=O)N[C@H](CO)[C@@H](C)O)C2=O)C1=O |r,t:17,19| |
|---|
| Structure | 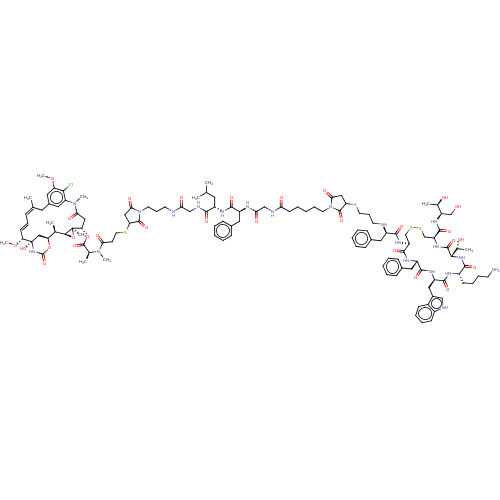
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 White, BH; Whalen, K; Kriksciukaite, K; Alargova, R; Au Yeung, T; Bazinet, P; Brockman, A; DuPont, M; Oller, H; Lemelin, CA; Lim Soo, P; Moreau, B; Perino, S; Quinn, JM; Sharma, G; Shinde, R; Sweryda-Krawiec, B; Wooster, R; Bilodeau, MT Discovery of an SSTR2-Targeting Maytansinoid Conjugate (PEN-221) with Potent Activity in Vitro and in Vivo. J Med Chem62:2708-2719 (2019) [PubMed] Article
White, BH; Whalen, K; Kriksciukaite, K; Alargova, R; Au Yeung, T; Bazinet, P; Brockman, A; DuPont, M; Oller, H; Lemelin, CA; Lim Soo, P; Moreau, B; Perino, S; Quinn, JM; Sharma, G; Shinde, R; Sweryda-Krawiec, B; Wooster, R; Bilodeau, MT Discovery of an SSTR2-Targeting Maytansinoid Conjugate (PEN-221) with Potent Activity in Vitro and in Vivo. J Med Chem62:2708-2719 (2019) [PubMed] Article