| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Serine/threonine-protein kinase LMTK3 |
|---|
| Ligand | BDBM158154 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1986602 (CHEMBL4620149) |
|---|
| IC50 | 4653±n/a nM |
|---|
| Citation |  Ortiz, MA; Michaels, H; Molina, B; Toenjes, S; Davis, J; Marconi, GD; Hecht, D; Gustafson, JL; Piedrafita, FJ; Nefzi, A Discovery of cyclic guanidine-linked sulfonamides as inhibitors of LMTK3 kinase. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article Ortiz, MA; Michaels, H; Molina, B; Toenjes, S; Davis, J; Marconi, GD; Hecht, D; Gustafson, JL; Piedrafita, FJ; Nefzi, A Discovery of cyclic guanidine-linked sulfonamides as inhibitors of LMTK3 kinase. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Serine/threonine-protein kinase LMTK3 |
|---|
| Name: | Serine/threonine-protein kinase LMTK3 |
|---|
| Synonyms: | 2.7.11.1 | KIAA1883 | LMTK3 | LMTK3_HUMAN | Lemur tyrosine kinase 3 | Serine/threonine-protein kinase LMTK3 | TYKLM3 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 153597.90 |
|---|
| Organism: | Homo sapiens |
|---|
| Description: | ChEMBL_119182 |
|---|
| Residue: | 1460 |
|---|
| Sequence: | MPAPGALILLAAVSASGCLASPAHPDGFALGRAPLAPPYAVVLISCSGLLAFIFLLLTCL
CCKRGDVGFKEFENPEGEDCSGEYTPPAEETSSSQSLPDVYILPLAEVSLPMPAPQPSHS
DMTTPLGLSRQHLSYLQEIGSGWFGKVILGEIFSDYTPAQVVVKELRASAGPLEQRKFIS
EAQPYRSLQHPNVLQCLGLCVETLPFLLIMEFCQLGDLKRYLRAQRPPEGLSPELPPRDL
RTLQRMGLEIARGLAHLHSHNYVHSDLALRNCLLTSDLTVRIGDYGLAHSNYKEDYYLTP
ERLWIPLRWAAPELLGELHGTFMVVDQSRESNIWSLGVTLWELFEFGAQPYRHLSDEEVL
AFVVRQQHVKLARPRLKLPYADYWYDILQSCWRPPAQRPSASDLQLQLTYLLSERPPRPP
PPPPPPRDGPFPWPWPPAHSAPRPGTLSSPFPLLDGFPGADPDDVLTVTESSRGLNLECL
WEKARRGAGRGGGAPAWQPASAPPAPHANPSNPFYEALSTPSVLPVISARSPSVSSEYYI
RLEEHGSPPEPLFPNDWDPLDPGVPAPQAPQAPSEVPQLVSETWASPLFPAPRPFPAQSS
ASGSFLLSGWDPEGRGAGETLAGDPAEVLGERGTAPWVEEEEEEEEGSSPGEDSSSLGGG
PSRRGPLPCPLCSREGACSCLPLERGDAVAGWGGHPALGCPHPPEDDSSLRAERGSLADL
PMAPPASAPPEFLDPLMGAAAPQYPGRGPPPAPPPPPPPPRAPADPAASPDPPSAVASPG
SGLSSPGPKPGDSGYETETPFSPEGAFPGGGAAEEEGVPRPRAPPEPPDPGAPRPPPDPG
PLPLPGPREKPTFVVQVSTEQLLMSLREDVTRNLLGEKGATARETGPRKAGRGPGNREKV
PGLNRDPTVLGNGKQAPSLSLPVNGVTVLENGDQRAPGIEEKAAENGALGSPEREEKVLE
NGELTPPRREEKALENGELRSPEAGEKVLVNGGLTPPKSEDKVSENGGLRFPRNTERPPE
TGPWRAPGPWEKTPESWGPAPTIGEPAPETSLERAPAPSAVVSSRNGGETAPGPLGPAPK
NGTLEPGTERRAPETGGAPRAPGAGRLDLGSGGRAPVGTGTAPGGGPGSGVDAKAGWVDN
TRPQPPPPPLPPPPEAQPRRLEPAPPRARPEVAPEGEPGAPDSRAGGDTALSGDGDPPKP
ERKGPEMPRLFLDLGPPQGNSEQIKARLSRLSLALPPLTLTPFPGPGPRRPPWEGADAGA
AGGEAGGAGAPGPAEEDGEDEDEDEEEDEEAAAPGAAAGPRGPGRARAAPVPVVVSSADA
DAARPLRGLLKSPRGADEPEDSELERKRKMVSFHGDVTVYLFDQETPTNELSVQAPPEGD
TDPSTPPAPPTPPHPATPGDGFPSNDSGFGGSFEWAEDFPLLPPPGPPLCFSRFSVSPAL
ETPGPPARAPDARPAGPVEN
|
|
|
|---|
| BDBM158154 |
|---|
| n/a |
|---|
| Name | BDBM158154 |
|---|
| Synonyms: | US10081622, Compound 11 | US10370379, Entrectinib | US11059827, Compound Entrectinib | US11267806, Compound T-12 | US9029356, 11 | US9255087, 11 | US9616059, 11 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H34F2N6O2 |
|---|
| Mol. Mass. | 560.6375 |
|---|
| SMILES | CN1CCN(CC1)c1ccc(C(=O)Nc2n[nH]c3ccc(Cc4cc(F)cc(F)c4)cc23)c(NC2CCOCC2)c1 |
|---|
| Structure | 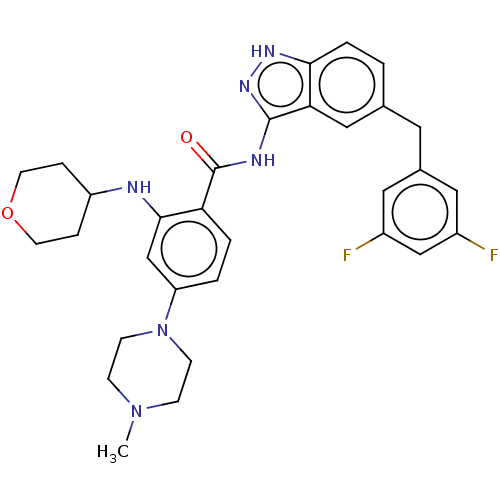
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ortiz, MA; Michaels, H; Molina, B; Toenjes, S; Davis, J; Marconi, GD; Hecht, D; Gustafson, JL; Piedrafita, FJ; Nefzi, A Discovery of cyclic guanidine-linked sulfonamides as inhibitors of LMTK3 kinase. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article
Ortiz, MA; Michaels, H; Molina, B; Toenjes, S; Davis, J; Marconi, GD; Hecht, D; Gustafson, JL; Piedrafita, FJ; Nefzi, A Discovery of cyclic guanidine-linked sulfonamides as inhibitors of LMTK3 kinase. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article