| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholinesterase |
|---|
| Ligand | BDBM50550233 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2027087 (CHEMBL4681245) |
|---|
| IC50 | 12±n/a nM |
|---|
| Citation |  Benchekroun, M; Romero, A; Egea, J; Le�n, R; Michalska, P; Buend�a, I; Jimeno, ML; Jun, D; Janockova, J; Sepsova, V; Soukup, O; Bautista-Aguilera, OM; Refouvelet, B; Ouari, O; Marco-Contelles, J; Ismaili, L The Antioxidant Additive Approach for Alzheimer's Disease Therapy: New Ferulic (Lipoic) Acid Plus Melatonin Modified Tacrines as Cholinesterases Inhibitors, Direct Antioxidants, and Nuclear Factor (Erythroid-Derived 2)-Like 2 Activators. J Med Chem59:9967-9973 (2016) [PubMed] Benchekroun, M; Romero, A; Egea, J; Le�n, R; Michalska, P; Buend�a, I; Jimeno, ML; Jun, D; Janockova, J; Sepsova, V; Soukup, O; Bautista-Aguilera, OM; Refouvelet, B; Ouari, O; Marco-Contelles, J; Ismaili, L The Antioxidant Additive Approach for Alzheimer's Disease Therapy: New Ferulic (Lipoic) Acid Plus Melatonin Modified Tacrines as Cholinesterases Inhibitors, Direct Antioxidants, and Nuclear Factor (Erythroid-Derived 2)-Like 2 Activators. J Med Chem59:9967-9973 (2016) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholinesterase |
|---|
| Name: | Cholinesterase |
|---|
| Synonyms: | BCHE | Butyrylcholinesterase (BuChE) | CHLE_HORSE | Cholinesterase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 65643.35 |
|---|
| Organism: | Equus caballus (Horse) |
|---|
| Description: | P81908 |
|---|
| Residue: | 574 |
|---|
| Sequence: | EEDIIITTKNGKVRGMNLPVLGGTVTAFLGIPYAQPPLGRLRFKKPQSLTKWSNIWNATK
YANSCYQNTDQSFPGFLGSEMWNPNTELSEDCLYLNVWIPAPKPKNATVMIWIYGGGFQT
GTSSLPVYDGKFLARVERVIVVSMNYRVGALGFLALSENPEAPGNMGLFDQQLALQWVQK
NIAAFGGNPRSVTLFGESAGAASVSLHLLSPRSQPLFTRAILQSGSSNAPWAVTSLYEAR
NRTLTLAKRMGCSRDNETEMIKCLRDKDPQEILLNEVFVVPYDTLLSVNFGPTVDGDFLT
DMPDTLLQLGQFKRTQILVGVNKDEGTAFLVYGAPGFSKDNNSIITRKEFQEGLKIFFPR
VSEFGRESILFHYMDWLDDQRAENYREALDDVVGDYNIICPALEFTRKFSELGNDAFFYY
FEHRSTKLPWPEWMGVMHGYEIEFVFGLPLERRVNYTRAEEILSRSIMKRWANFAKYGNP
NGTQNNSTRWPVFKSTEQKYLTLNTESPKVYTKLRAQQCRFWTLFFPKVLELTGNIDEAE
REWKAGFHRWNNYMMDWKNQFNDYTSKKESCSDF
|
|
|
|---|
| BDBM50550233 |
|---|
| n/a |
|---|
| Name | BDBM50550233 |
|---|
| Synonyms: | CHEMBL4762095 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C45H55N5O6 |
|---|
| Mol. Mass. | 761.9481 |
|---|
| SMILES | COc1ccc2[nH]cc(CCNC(=O)CN(CCCCCCCCNc3c4CCCCc4nc4ccc(OC)cc34)C(=O)\C=C\c3ccc(O)c(OC)c3)c2c1 |
|---|
| Structure | 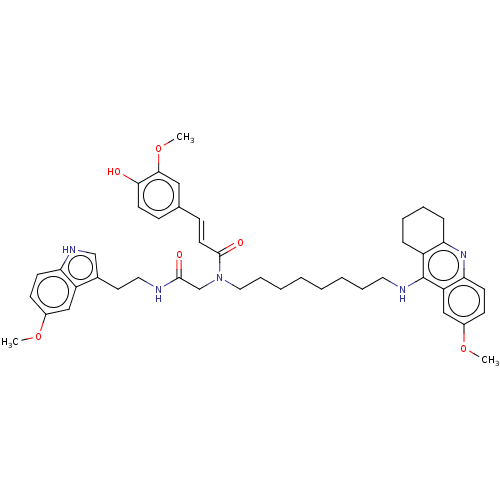
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Benchekroun, M; Romero, A; Egea, J; Le�n, R; Michalska, P; Buend�a, I; Jimeno, ML; Jun, D; Janockova, J; Sepsova, V; Soukup, O; Bautista-Aguilera, OM; Refouvelet, B; Ouari, O; Marco-Contelles, J; Ismaili, L The Antioxidant Additive Approach for Alzheimer's Disease Therapy: New Ferulic (Lipoic) Acid Plus Melatonin Modified Tacrines as Cholinesterases Inhibitors, Direct Antioxidants, and Nuclear Factor (Erythroid-Derived 2)-Like 2 Activators. J Med Chem59:9967-9973 (2016) [PubMed]
Benchekroun, M; Romero, A; Egea, J; Le�n, R; Michalska, P; Buend�a, I; Jimeno, ML; Jun, D; Janockova, J; Sepsova, V; Soukup, O; Bautista-Aguilera, OM; Refouvelet, B; Ouari, O; Marco-Contelles, J; Ismaili, L The Antioxidant Additive Approach for Alzheimer's Disease Therapy: New Ferulic (Lipoic) Acid Plus Melatonin Modified Tacrines as Cholinesterases Inhibitors, Direct Antioxidants, and Nuclear Factor (Erythroid-Derived 2)-Like 2 Activators. J Med Chem59:9967-9973 (2016) [PubMed]