| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50554717 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2050396 (CHEMBL4705095) |
|---|
| IC50 | 3500±n/a nM |
|---|
| Citation |  Furuya, T; Shapiro, AB; Comita-Prevoir, J; Kuenstner, EJ; Zhang, J; Ribe, SD; Chen, A; Hines, D; Moussa, SH; Carter, NM; Sylvester, MA; Romero, JAC; Vega, CV; Sacco, MD; Chen, Y; O'Donnell, JP; Durand-Reville, TF; Miller, AA; Tommasi, RA N-Hydroxyformamide LpxC inhibitors, their in vivo efficacy in a mouse Escherichia coli infection model, and their safety in a rat hemodynamic assay. Bioorg Med Chem28:0 (2020) [PubMed] Article Furuya, T; Shapiro, AB; Comita-Prevoir, J; Kuenstner, EJ; Zhang, J; Ribe, SD; Chen, A; Hines, D; Moussa, SH; Carter, NM; Sylvester, MA; Romero, JAC; Vega, CV; Sacco, MD; Chen, Y; O'Donnell, JP; Durand-Reville, TF; Miller, AA; Tommasi, RA N-Hydroxyformamide LpxC inhibitors, their in vivo efficacy in a mouse Escherichia coli infection model, and their safety in a rat hemodynamic assay. Bioorg Med Chem28:0 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_HUMAN | ACHE | Acetylcholinesterase (AChE) | Acetylcholinesterase (human AChE) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67792.70 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22303 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPV
SAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSM
NYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASV
GMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTEL
VACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVG
VVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPE
DPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGY
EIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQ
YVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQDRCSDL
|
|
|
|---|
| BDBM50554717 |
|---|
| n/a |
|---|
| Name | BDBM50554717 |
|---|
| Synonyms: | CHEMBL4746231 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H25N5O3S |
|---|
| Mol. Mass. | 451.541 |
|---|
| SMILES | CS[C@@H](Cn1nnc2cc(ccc12)C#Cc1ccc(CN2CCOCC2)cc1)N(O)C=O |r| |
|---|
| Structure | 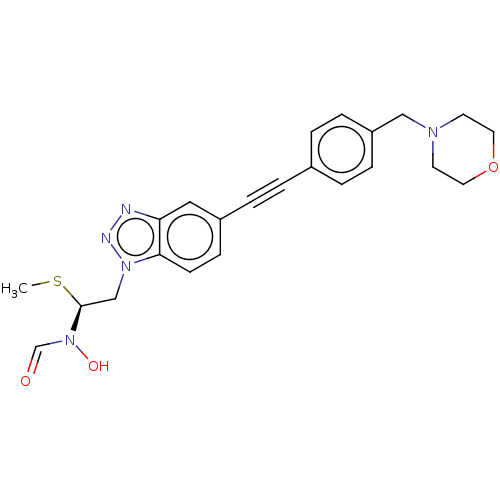
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Furuya, T; Shapiro, AB; Comita-Prevoir, J; Kuenstner, EJ; Zhang, J; Ribe, SD; Chen, A; Hines, D; Moussa, SH; Carter, NM; Sylvester, MA; Romero, JAC; Vega, CV; Sacco, MD; Chen, Y; O'Donnell, JP; Durand-Reville, TF; Miller, AA; Tommasi, RA N-Hydroxyformamide LpxC inhibitors, their in vivo efficacy in a mouse Escherichia coli infection model, and their safety in a rat hemodynamic assay. Bioorg Med Chem28:0 (2020) [PubMed] Article
Furuya, T; Shapiro, AB; Comita-Prevoir, J; Kuenstner, EJ; Zhang, J; Ribe, SD; Chen, A; Hines, D; Moussa, SH; Carter, NM; Sylvester, MA; Romero, JAC; Vega, CV; Sacco, MD; Chen, Y; O'Donnell, JP; Durand-Reville, TF; Miller, AA; Tommasi, RA N-Hydroxyformamide LpxC inhibitors, their in vivo efficacy in a mouse Escherichia coli infection model, and their safety in a rat hemodynamic assay. Bioorg Med Chem28:0 (2020) [PubMed] Article