| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor alpha |
|---|
| Ligand | BDBM50148098 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_153534 (CHEMBL765403) |
|---|
| IC50 | >15000±n/a nM |
|---|
| Citation |  Koyama, H; Miller, DJ; Boueres, JK; Desai, RC; Jones, AB; Berger, JP; MacNaul, KL; Kelly, LJ; Doebber, TW; Wu, MS; Zhou, G; Wang, PR; Ippolito, MC; Chao, YS; Agrawal, AK; Franklin, R; Heck, JV; Wright, SD; Moller, DE; Sahoo, SP (2R)-2-ethylchromane-2-carboxylic acids: discovery of novel PPARalpha/gamma dual agonists as antihyperglycemic and hypolipidemic agents. J Med Chem47:3255-63 (2004) [PubMed] Article Koyama, H; Miller, DJ; Boueres, JK; Desai, RC; Jones, AB; Berger, JP; MacNaul, KL; Kelly, LJ; Doebber, TW; Wu, MS; Zhou, G; Wang, PR; Ippolito, MC; Chao, YS; Agrawal, AK; Franklin, R; Heck, JV; Wright, SD; Moller, DE; Sahoo, SP (2R)-2-ethylchromane-2-carboxylic acids: discovery of novel PPARalpha/gamma dual agonists as antihyperglycemic and hypolipidemic agents. J Med Chem47:3255-63 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor alpha |
|---|
| Name: | Peroxisome proliferator-activated receptor alpha |
|---|
| Synonyms: | NR1C1 | Nuclear receptor subfamily 1 group C member 1 | PPAR | PPAR alpha/gamma | PPAR-alpha | PPARA | PPARA_HUMAN | Peroxisome Proliferator-Activated Receptor alpha | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor alpha (PPAR alpha) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 52222.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q07869 |
|---|
| Residue: | 468 |
|---|
| Sequence: | MVDTESPLCPLSPLEAGDLESPLSEEFLQEMGNIQEISQSIGEDSSGSFGFTEYQYLGSC
PGSDGSVITDTLSPASSPSSVTYPVVPGSVDESPSGALNIECRICGDKASGYHYGVHACE
GCKGFFRRTIRLKLVYDKCDRSCKIQKKNRNKCQYCRFHKCLSVGMSHNAIRFGRMPRSE
KAKLKAEILTCEHDIEDSETADLKSLAKRIYEAYLKNFNMNKVKARVILSGKASNNPPFV
IHDMETLCMAEKTLVAKLVANGIQNKEAEVRIFHCCQCTSVETVTELTEFAKAIPGFANL
DLNDQVTLLKYGVYEAIFAMLSSVMNKDGMLVAYGNGFITREFLKSLRKPFCDIMEPKFD
FAMKFNALELDDSDISLFVAAIICCGDRPGLLNVGHIEKMQEGIVHVLRLHLQSNHPDDI
FLFPKLLQKMADLRQLVTEHAQLVQIIKKTESDAALHPLLQEIYRDMY
|
|
|
|---|
| BDBM50148098 |
|---|
| n/a |
|---|
| Name | BDBM50148098 |
|---|
| Synonyms: | (S)-7-[3-(2-Chloro-4-phenoxy-phenoxy)-propoxy]-2-methyl-chroman-2-carboxylic acid | CHEMBL112790 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H25ClO6 |
|---|
| Mol. Mass. | 468.926 |
|---|
| SMILES | C[C@]1(CCc2ccc(OCCCOc3ccc(Oc4ccccc4)cc3Cl)cc2O1)C(O)=O |
|---|
| Structure | 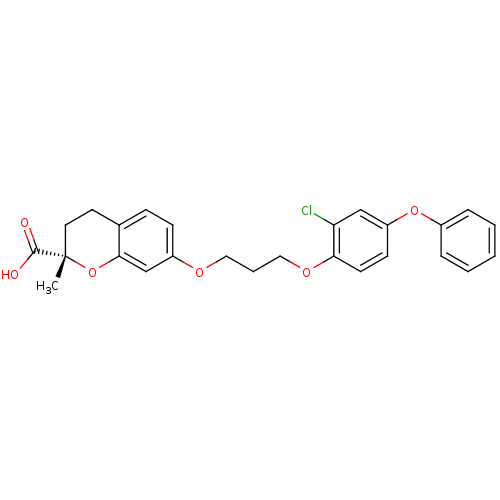
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Koyama, H; Miller, DJ; Boueres, JK; Desai, RC; Jones, AB; Berger, JP; MacNaul, KL; Kelly, LJ; Doebber, TW; Wu, MS; Zhou, G; Wang, PR; Ippolito, MC; Chao, YS; Agrawal, AK; Franklin, R; Heck, JV; Wright, SD; Moller, DE; Sahoo, SP (2R)-2-ethylchromane-2-carboxylic acids: discovery of novel PPARalpha/gamma dual agonists as antihyperglycemic and hypolipidemic agents. J Med Chem47:3255-63 (2004) [PubMed] Article
Koyama, H; Miller, DJ; Boueres, JK; Desai, RC; Jones, AB; Berger, JP; MacNaul, KL; Kelly, LJ; Doebber, TW; Wu, MS; Zhou, G; Wang, PR; Ippolito, MC; Chao, YS; Agrawal, AK; Franklin, R; Heck, JV; Wright, SD; Moller, DE; Sahoo, SP (2R)-2-ethylchromane-2-carboxylic acids: discovery of novel PPARalpha/gamma dual agonists as antihyperglycemic and hypolipidemic agents. J Med Chem47:3255-63 (2004) [PubMed] Article