| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sphingosine 1-phosphate receptor 2 |
|---|
| Ligand | BDBM50585280 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2161846 (CHEMBL5046707) |
|---|
| IC50 | 3.4±n/a nM |
|---|
| Citation |  Mammoliti, O; Jansen, K; El Bkassiny, S; Palisse, A; Triballeau, N; Bucher, D; Allart, B; Jaunet, A; Tricarico, G; De Wachter, M; Menet, C; Blanc, J; Letfus, V; Rup?i?, R; ?mehil, M; Poljak, T; Coornaert, B; Sonck, K; Duys, I; Waeckel, L; Lecru, L; Marsais, F; Jagerschmidt, C; Auberval, M; Pujuguet, P; Oste, L; Borgonovi, M; Wakselman, E; Christophe, T; Houvenaghel, N; Jans, M; Heckmann, B; Sani�re, L; Brys, R Discovery and Optimization of Orally Bioavailable Phthalazone and Cinnolone Carboxylic Acid Derivatives as S1P2 Antagonists against Fibrotic Diseases. J Med Chem64:14557-14586 (2021) [PubMed] Article Mammoliti, O; Jansen, K; El Bkassiny, S; Palisse, A; Triballeau, N; Bucher, D; Allart, B; Jaunet, A; Tricarico, G; De Wachter, M; Menet, C; Blanc, J; Letfus, V; Rup?i?, R; ?mehil, M; Poljak, T; Coornaert, B; Sonck, K; Duys, I; Waeckel, L; Lecru, L; Marsais, F; Jagerschmidt, C; Auberval, M; Pujuguet, P; Oste, L; Borgonovi, M; Wakselman, E; Christophe, T; Houvenaghel, N; Jans, M; Heckmann, B; Sani�re, L; Brys, R Discovery and Optimization of Orally Bioavailable Phthalazone and Cinnolone Carboxylic Acid Derivatives as S1P2 Antagonists against Fibrotic Diseases. J Med Chem64:14557-14586 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sphingosine 1-phosphate receptor 2 |
|---|
| Name: | Sphingosine 1-phosphate receptor 2 |
|---|
| Synonyms: | EDG5 | S1P2 | S1PR2 | S1PR2_HUMAN | Sphingosine 1-phosphate receptor | Sphingosine 1-phosphate receptor Edg-5 | Sphingosine-1-phosphate receptor 2 | ndothelial differentiation G-protein coupled receptor 5 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 38883.16 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Membranes isolated from S1P2-transfected CHO cells were used in ligand binding assay. |
|---|
| Residue: | 353 |
|---|
| Sequence: | MGSLYSEYLNPNKVQEHYNYTKETLETQETTSRQVASAFIVILCCAIVVENLLVLIAVAR
NSKFHSAMYLFLGNLAASDLLAGVAFVANTLLSGSVTLRLTPVQWFAREGSAFITLSASV
FSLLAIAIERHVAIAKVKLYGSDKSCRMLLLIGASWLISLVLGGLPILGWNCLGHLEACS
TVLPLYAKHYVLCVVTIFSIILLAIVALYVRIYCVVRSSHADMAAPQTLALLKTVTIVLG
VFIVCWLPAFSILLLDYACPVHSCPILYKAHYFFAVSTLNSLLNPVIYTWRSRDLRREVL
RPLQCWRPGVGVQGRRRGGTPGHHLLPLRSSSSLERGMHMPTSPTFLEGNTVV
|
|
|
|---|
| BDBM50585280 |
|---|
| n/a |
|---|
| Name | BDBM50585280 |
|---|
| Synonyms: | CHEMBL5090966 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H21F3N4O4 |
|---|
| Mol. Mass. | 486.4431 |
|---|
| SMILES | CC(C(O)=O)c1nn(Cc2cn3cc(OCC4CC4)c(cc3n2)C(F)(F)F)c2ccccc2c1=O |
|---|
| Structure | 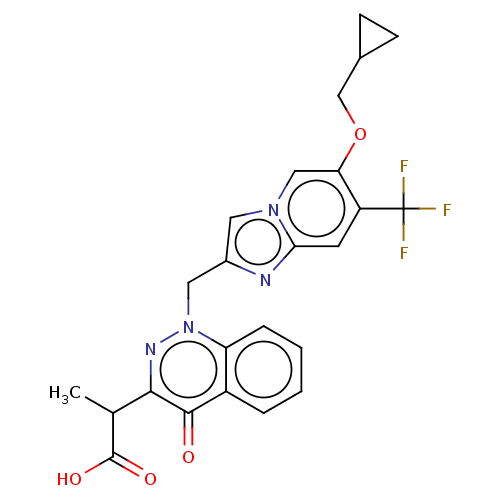
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mammoliti, O; Jansen, K; El Bkassiny, S; Palisse, A; Triballeau, N; Bucher, D; Allart, B; Jaunet, A; Tricarico, G; De Wachter, M; Menet, C; Blanc, J; Letfus, V; Rup?i?, R; ?mehil, M; Poljak, T; Coornaert, B; Sonck, K; Duys, I; Waeckel, L; Lecru, L; Marsais, F; Jagerschmidt, C; Auberval, M; Pujuguet, P; Oste, L; Borgonovi, M; Wakselman, E; Christophe, T; Houvenaghel, N; Jans, M; Heckmann, B; Sani�re, L; Brys, R Discovery and Optimization of Orally Bioavailable Phthalazone and Cinnolone Carboxylic Acid Derivatives as S1P2 Antagonists against Fibrotic Diseases. J Med Chem64:14557-14586 (2021) [PubMed] Article
Mammoliti, O; Jansen, K; El Bkassiny, S; Palisse, A; Triballeau, N; Bucher, D; Allart, B; Jaunet, A; Tricarico, G; De Wachter, M; Menet, C; Blanc, J; Letfus, V; Rup?i?, R; ?mehil, M; Poljak, T; Coornaert, B; Sonck, K; Duys, I; Waeckel, L; Lecru, L; Marsais, F; Jagerschmidt, C; Auberval, M; Pujuguet, P; Oste, L; Borgonovi, M; Wakselman, E; Christophe, T; Houvenaghel, N; Jans, M; Heckmann, B; Sani�re, L; Brys, R Discovery and Optimization of Orally Bioavailable Phthalazone and Cinnolone Carboxylic Acid Derivatives as S1P2 Antagonists against Fibrotic Diseases. J Med Chem64:14557-14586 (2021) [PubMed] Article