| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cysteinyl leukotriene receptor 1 |
|---|
| Ligand | BDBM50009073 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2219705 (CHEMBL5133039) |
|---|
| IC50 | 0.260000±n/a nM |
|---|
| Citation |  Fiorillo, B; Sepe, V; Conflitti, P; Roselli, R; Biagioli, M; Marchian�, S; De Luca, P; Baronissi, G; Rapacciuolo, P; Cassiano, C; Catalanotti, B; Zampella, A; Limongelli, V; Fiorucci, S Structural Basis for Developing Multitarget Compounds Acting on Cysteinyl Leukotriene Receptor 1 and G-Protein-Coupled Bile Acid Receptor 1. J Med Chem64:16512-16529 (2021) [PubMed] Article Fiorillo, B; Sepe, V; Conflitti, P; Roselli, R; Biagioli, M; Marchian�, S; De Luca, P; Baronissi, G; Rapacciuolo, P; Cassiano, C; Catalanotti, B; Zampella, A; Limongelli, V; Fiorucci, S Structural Basis for Developing Multitarget Compounds Acting on Cysteinyl Leukotriene Receptor 1 and G-Protein-Coupled Bile Acid Receptor 1. J Med Chem64:16512-16529 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cysteinyl leukotriene receptor 1 |
|---|
| Name: | Cysteinyl leukotriene receptor 1 |
|---|
| Synonyms: | CLTR1_HUMAN | CYSLT1 | CYSLTR1 | Cysteinyl leukotriene D4 receptor | Cysteinyl leukotriene receptor | Cysteinyl leukotriene receptor 1 | HG55 | HMTMF81 | LTD4 receptor | Leukotriene Cysteinyl 1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 38565.16 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Leukotriene Cysteinyl 1 CYSLTR1 HUMAN::Q9Y271 |
|---|
| Residue: | 337 |
|---|
| Sequence: | MDETGNLTVSSATCHDTIDDFRNQVYSTLYSMISVVGFFGNGFVLYVLIKTYHKKSAFQV
YMINLAVADLLCVCTLPLRVVYYVHKGIWLFGDFLCRLSTYALYVNLYCSIFFMTAMSFF
RCIAIVFPVQNINLVTQKKARFVCVGIWIFVILTSSPFLMAKPQKDEKNNTKCFEPPQDN
QTKNHVLVLHYVSLFVGFIIPFVIIIVCYTMIILTLLKKSMKKNLSSHKKAIGMIMVVTA
AFLVSFMPYHIQRTIHLHFLHNETKPCDSVLRMQKSVVITLSLAASNCCFDPLLYFFSGG
NFRKRLSTFRKHSLSSVTYVPRKKASLPEKGEEICKV
|
|
|
|---|
| BDBM50009073 |
|---|
| n/a |
|---|
| Name | BDBM50009073 |
|---|
| Synonyms: | 4-(5-cyclopentyloxycarbonylamino-1-methyl-1H-indol-3-ylmethyl)-3-methoxy-N-o-tolylsulfonylbenzamide | CHEMBL603 | ZAFIRLUKAST | cyclopentyl 3-(2-methoxy-4-((o-tolylsulfonyl)carbamoyl)benzyl)-1-methylindole-5-carbamate | cyclopentyl 3-[2-methoxy-4-(2-methylphenylsulfonylcarbamoyl)benzyl]-1-methyl-1H-indol-5-ylcarbamate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H33N3O6S |
|---|
| Mol. Mass. | 575.675 |
|---|
| SMILES | COc1cc(ccc1Cc1cn(C)c2ccc(NC(=O)OC3CCCC3)cc12)C(=O)NS(=O)(=O)c1ccccc1C |
|---|
| Structure | 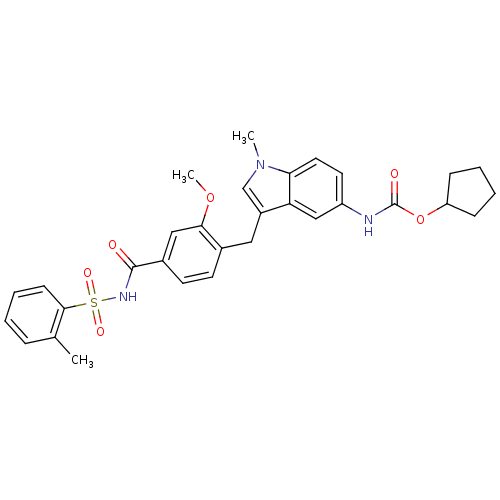
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fiorillo, B; Sepe, V; Conflitti, P; Roselli, R; Biagioli, M; Marchian�, S; De Luca, P; Baronissi, G; Rapacciuolo, P; Cassiano, C; Catalanotti, B; Zampella, A; Limongelli, V; Fiorucci, S Structural Basis for Developing Multitarget Compounds Acting on Cysteinyl Leukotriene Receptor 1 and G-Protein-Coupled Bile Acid Receptor 1. J Med Chem64:16512-16529 (2021) [PubMed] Article
Fiorillo, B; Sepe, V; Conflitti, P; Roselli, R; Biagioli, M; Marchian�, S; De Luca, P; Baronissi, G; Rapacciuolo, P; Cassiano, C; Catalanotti, B; Zampella, A; Limongelli, V; Fiorucci, S Structural Basis for Developing Multitarget Compounds Acting on Cysteinyl Leukotriene Receptor 1 and G-Protein-Coupled Bile Acid Receptor 1. J Med Chem64:16512-16529 (2021) [PubMed] Article