| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Calcitonin gene-related peptide type 1 receptor |
|---|
| Ligand | BDBM50173468 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_321551 (CHEMBL881965) |
|---|
| pH | 7.4±n/a |
|---|
| IC50 | 6.4±n/a nM |
|---|
| Comments | extracted |
|---|
| Citation |  Rudolf, K; Eberlein, W; Engel, W; Pieper, H; Entzeroth, M; Hallermayer, G; Doods, H Development of human calcitonin gene-related peptide (CGRP) receptor antagonists. 1. Potent and selective small molecule CGRP antagonists. 1-[N2-[3,5-dibromo-N-[[4-(3,4-dihydro-2(1H)-oxoquinazolin-3-yl)-1-piperidinyl]carbonyl]-D-tyrosyl]-l-lysyl]-4-(4-pyridinyl)piperazine: the first CGRP antagonist J Med Chem48:5921-31 (2005) [PubMed] Article Rudolf, K; Eberlein, W; Engel, W; Pieper, H; Entzeroth, M; Hallermayer, G; Doods, H Development of human calcitonin gene-related peptide (CGRP) receptor antagonists. 1. Potent and selective small molecule CGRP antagonists. 1-[N2-[3,5-dibromo-N-[[4-(3,4-dihydro-2(1H)-oxoquinazolin-3-yl)-1-piperidinyl]carbonyl]-D-tyrosyl]-l-lysyl]-4-(4-pyridinyl)piperazine: the first CGRP antagonist J Med Chem48:5921-31 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Calcitonin gene-related peptide type 1 receptor |
|---|
| Name: | Calcitonin gene-related peptide type 1 receptor |
|---|
| Synonyms: | CALRL_RAT | Calcitonin-gene-related peptide receptor; CALCRL/RAMP1 | Calcrl | Cgrpr |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 53330.91 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_1457286 |
|---|
| Residue: | 464 |
|---|
| Sequence: | MMDKKCTLCFLFLLLLNMALIAAESEEGANQTDLGVTRNKIMTAQYECYQKIMQDPIQQG
EGLYCNRTWDGWLCWNDVAAGTESMQYCPDYFQDFDPSEKVTKICDQDGNWFRHPDSNRT
WTNYTLCNNSTHEKVKTALNLFYLTIIGHGLSIASLIISLIIFFYFKSLSCQRITLHKNL
FFSFVCNSIVTIIHLTAVANNQALVATNPVSCKVSQFIHLYLMGCNYFWMLCEGIYLHTL
IVVAVFAEKQHLMWYYFLGWGFPLLPACIHAIARSLYYNDNCWISSDTHLLYIIHGPICA
ALLVNLFFLLNIVRVLITKLKVTHQAESNLYMKAVRATLILVPLLGIEFVLFPWRPEGKV
AEEVYDYVMHILMHYQGLLVSTIFCFFNGEVQAILRRNWNQYKIQFGNGFSHSDALRSAS
YTVSTISDVQGYSHDCPTEHLNGKSIQDIENVALKPEKMYDLVM
|
|
|
|---|
| BDBM50173468 |
|---|
| n/a |
|---|
| Name | BDBM50173468 |
|---|
| Synonyms: | 4-(2-Oxo-1,4-dihydro-2H-quinazolin-3-yl)-piperidine-1-carboxylic acid [(S)-1-[(R)-5-amino-1-(4-pyridin-4-yl-piperazine-1-carbonyl)-pentylcarbamoyl]-2-(3,5-dibromo-4-hydroxy-phenyl)-ethyl]-amide | CHEMBL197529 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C38H47Br2N9O5 |
|---|
| Mol. Mass. | 869.645 |
|---|
| SMILES | NCCCC[C@@H](NC(=O)[C@H](Cc1cc(Br)c(O)c(Br)c1)NC(=O)N1CCC(CC1)N1Cc2ccccc2NC1=O)C(=O)N1CCN(CC1)c1ccncc1 |
|---|
| Structure | 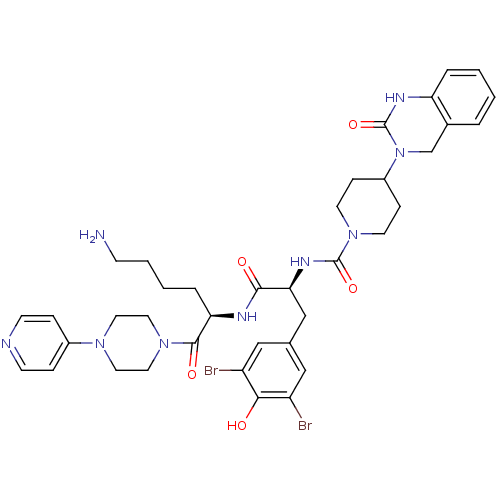
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Rudolf, K; Eberlein, W; Engel, W; Pieper, H; Entzeroth, M; Hallermayer, G; Doods, H Development of human calcitonin gene-related peptide (CGRP) receptor antagonists. 1. Potent and selective small molecule CGRP antagonists. 1-[N2-[3,5-dibromo-N-[[4-(3,4-dihydro-2(1H)-oxoquinazolin-3-yl)-1-piperidinyl]carbonyl]-D-tyrosyl]-l-lysyl]-4-(4-pyridinyl)piperazine: the first CGRP antagonist J Med Chem48:5921-31 (2005) [PubMed] Article
Rudolf, K; Eberlein, W; Engel, W; Pieper, H; Entzeroth, M; Hallermayer, G; Doods, H Development of human calcitonin gene-related peptide (CGRP) receptor antagonists. 1. Potent and selective small molecule CGRP antagonists. 1-[N2-[3,5-dibromo-N-[[4-(3,4-dihydro-2(1H)-oxoquinazolin-3-yl)-1-piperidinyl]carbonyl]-D-tyrosyl]-l-lysyl]-4-(4-pyridinyl)piperazine: the first CGRP antagonist J Med Chem48:5921-31 (2005) [PubMed] Article