| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50391283 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2264099 |
|---|
| IC50 | >45000±n/a nM |
|---|
| Citation |  Nirogi, R; Mohammed, AR; Shinde, AK; Gagginapally, SR; Kancharla, DM; Ravella, SR; Bogaraju, N; Middekadi, VR; Subramanian, R; Palacharla, RC; Benade, V; Muddana, N; Abraham, R; Medapati, RB; Thentu, JB; Mekala, VR; Petlu, S; Lingavarapu, BB; Yarra, S; Kagita, N; Goyal, VK; Pandey, SK; Jasti, V Discovery and Preclinical Characterization of Usmarapride (SUVN-D4010): A Potent, Selective 5-HT J Med Chem64:10641-10665 (2021) [PubMed] Nirogi, R; Mohammed, AR; Shinde, AK; Gagginapally, SR; Kancharla, DM; Ravella, SR; Bogaraju, N; Middekadi, VR; Subramanian, R; Palacharla, RC; Benade, V; Muddana, N; Abraham, R; Medapati, RB; Thentu, JB; Mekala, VR; Petlu, S; Lingavarapu, BB; Yarra, S; Kagita, N; Goyal, VK; Pandey, SK; Jasti, V Discovery and Preclinical Characterization of Usmarapride (SUVN-D4010): A Potent, Selective 5-HT J Med Chem64:10641-10665 (2021) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50391283 |
|---|
| n/a |
|---|
| Name | BDBM50391283 |
|---|
| Synonyms: | CHEMBL5268064 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H29N5O6 |
|---|
| Mol. Mass. | 459.4956 |
|---|
| SMILES | OC(=O)C(O)=O.CC(C)n1nc(-c2nnc(o2)C2CCN(CCCO)CC2)c2ccccc12 |
|---|
| Structure | 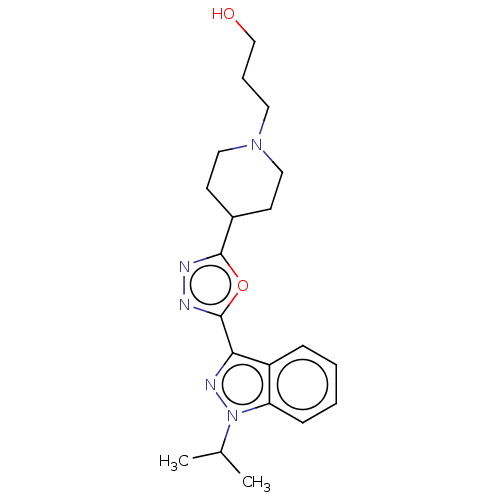
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nirogi, R; Mohammed, AR; Shinde, AK; Gagginapally, SR; Kancharla, DM; Ravella, SR; Bogaraju, N; Middekadi, VR; Subramanian, R; Palacharla, RC; Benade, V; Muddana, N; Abraham, R; Medapati, RB; Thentu, JB; Mekala, VR; Petlu, S; Lingavarapu, BB; Yarra, S; Kagita, N; Goyal, VK; Pandey, SK; Jasti, V Discovery and Preclinical Characterization of Usmarapride (SUVN-D4010): A Potent, Selective 5-HT J Med Chem64:10641-10665 (2021) [PubMed]
Nirogi, R; Mohammed, AR; Shinde, AK; Gagginapally, SR; Kancharla, DM; Ravella, SR; Bogaraju, N; Middekadi, VR; Subramanian, R; Palacharla, RC; Benade, V; Muddana, N; Abraham, R; Medapati, RB; Thentu, JB; Mekala, VR; Petlu, S; Lingavarapu, BB; Yarra, S; Kagita, N; Goyal, VK; Pandey, SK; Jasti, V Discovery and Preclinical Characterization of Usmarapride (SUVN-D4010): A Potent, Selective 5-HT J Med Chem64:10641-10665 (2021) [PubMed]