| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor gamma |
|---|
| Ligand | BDBM50610167 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2280298 |
|---|
| IC50 | 24±n/a nM |
|---|
| Citation |  Orsi, DL; Ferrara, SJ; Siegel, S; Friberg, A; Bouch�, L; Pook, E; Lienau, P; Bluck, JP; Lemke, CT; Akcay, G; Stellfeld, T; Meyer, H; P�tter, V; Holton, SJ; Korr, D; Jerchel-Furau, I; Pantelidou, C; Strathdee, CA; Meyerson, M; Eis, K; Goldstein, JT Discovery and characterization of orally bioavailable 4-chloro-6-fluoroisophthalamides as covalent PPARG inverse-agonists. Bioorg Med Chem78:0 (2023) [PubMed] Orsi, DL; Ferrara, SJ; Siegel, S; Friberg, A; Bouch�, L; Pook, E; Lienau, P; Bluck, JP; Lemke, CT; Akcay, G; Stellfeld, T; Meyer, H; P�tter, V; Holton, SJ; Korr, D; Jerchel-Furau, I; Pantelidou, C; Strathdee, CA; Meyerson, M; Eis, K; Goldstein, JT Discovery and characterization of orally bioavailable 4-chloro-6-fluoroisophthalamides as covalent PPARG inverse-agonists. Bioorg Med Chem78:0 (2023) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor gamma |
|---|
| Name: | Peroxisome proliferator-activated receptor gamma |
|---|
| Synonyms: | Nr1c3 | Nuclear receptor subfamily 1 group C member 3 | PPAR-gamma | PPARG_MOUSE | Pparg |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 57593.55 |
|---|
| Organism: | Mus musculus |
|---|
| Description: | ChEMBL_1453700 |
|---|
| Residue: | 505 |
|---|
| Sequence: | MGETLGDSPVDPEHGAFADALPMSTSQEITMVDTEMPFWPTNFGISSVDLSVMEDHSHSF
DIKPFTTVDFSSISAPHYEDIPFTRADPMVADYKYDLKLQEYQSAIKVEPASPPYYSEKT
QLYNRPHEEPSNSLMAIECRVCGDKASGFHYGVHACEGCKGFFRRTIRLKLIYDRCDLNC
RIHKKSRNKCQYCRFQKCLAVGMSHNAIRFGRMPQAEKEKLLAEISSDIDQLNPESADLR
ALAKHLYDSYIKSFPLTKAKARAILTGKTTDKSPFVIYDMNSLMMGEDKIKFKHITPLQE
QSKEVAIRIFQGCQFRSVEAVQEITEYAKNIPGFINLDLNDQVTLLKYGVHEIIYTMLAS
LMNKDGVLISEGQGFMTREFLKNLRKPFGDFMEPKFEFAVKFNALELDDSDLAIFIAVII
LSGDRPGLLNVKPIEDIQDNLLQALELQLKLNHPESSQLFAKVLQKMTDLRQIVTEHVQL
LHVIKKTETDMSLHPLLQEIYKDLY
|
|
|
|---|
| BDBM50610167 |
|---|
| n/a |
|---|
| Name | BDBM50610167 |
|---|
| Synonyms: | CHEMBL5271038 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H22ClF3N4O3 |
|---|
| Mol. Mass. | 518.915 |
|---|
| SMILES | Cc1cc(cnc1NC(=O)c1cc(C(=O)NCc2ccc(F)c(F)c2)c(F)cc1Cl)N1CCOCC1 |
|---|
| Structure | 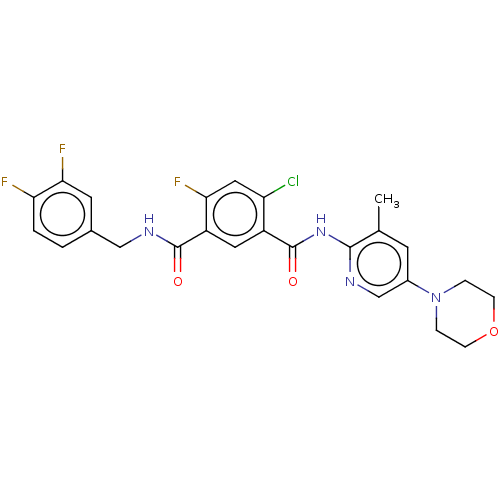
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Orsi, DL; Ferrara, SJ; Siegel, S; Friberg, A; Bouch�, L; Pook, E; Lienau, P; Bluck, JP; Lemke, CT; Akcay, G; Stellfeld, T; Meyer, H; P�tter, V; Holton, SJ; Korr, D; Jerchel-Furau, I; Pantelidou, C; Strathdee, CA; Meyerson, M; Eis, K; Goldstein, JT Discovery and characterization of orally bioavailable 4-chloro-6-fluoroisophthalamides as covalent PPARG inverse-agonists. Bioorg Med Chem78:0 (2023) [PubMed]
Orsi, DL; Ferrara, SJ; Siegel, S; Friberg, A; Bouch�, L; Pook, E; Lienau, P; Bluck, JP; Lemke, CT; Akcay, G; Stellfeld, T; Meyer, H; P�tter, V; Holton, SJ; Korr, D; Jerchel-Furau, I; Pantelidou, C; Strathdee, CA; Meyerson, M; Eis, K; Goldstein, JT Discovery and characterization of orally bioavailable 4-chloro-6-fluoroisophthalamides as covalent PPARG inverse-agonists. Bioorg Med Chem78:0 (2023) [PubMed]