| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50209925 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_443912 (CHEMBL893078) |
|---|
| IC50 | >50000±n/a nM |
|---|
| Citation |  Lübbers, T; Böhringer, M; Gobbi, L; Hennig, M; Hunziker, D; Kuhn, B; Löffler, B; Mattei, P; Narquizian, R; Peters, JU; Ruff, Y; Wessel, HP; Wyss, P 1,3-disubstituted 4-aminopiperidines as useful tools in the optimization of the 2-aminobenzo[a]quinolizine dipeptidyl peptidase IV inhibitors. Bioorg Med Chem Lett17:2966-70 (2007) [PubMed] Article Lübbers, T; Böhringer, M; Gobbi, L; Hennig, M; Hunziker, D; Kuhn, B; Löffler, B; Mattei, P; Narquizian, R; Peters, JU; Ruff, Y; Wessel, HP; Wyss, P 1,3-disubstituted 4-aminopiperidines as useful tools in the optimization of the 2-aminobenzo[a]quinolizine dipeptidyl peptidase IV inhibitors. Bioorg Med Chem Lett17:2966-70 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50209925 |
|---|
| n/a |
|---|
| Name | BDBM50209925 |
|---|
| Synonyms: | (2S,3S,11bS)-9,10-dimethoxy-3-m-tolyl-2,3,4,6,7,11b-hexahydro-1H-pyrido[2,1-a]isoquinolin-2-amine | CHEMBL391070 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H28N2O2 |
|---|
| Mol. Mass. | 352.4699 |
|---|
| SMILES | COc1cc2CCN3C[C@@H]([C@@H](N)C[C@H]3c2cc1OC)c1cccc(C)c1 |
|---|
| Structure | 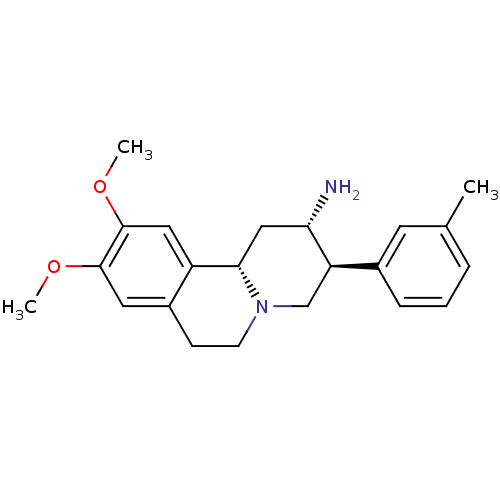
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lübbers, T; Böhringer, M; Gobbi, L; Hennig, M; Hunziker, D; Kuhn, B; Löffler, B; Mattei, P; Narquizian, R; Peters, JU; Ruff, Y; Wessel, HP; Wyss, P 1,3-disubstituted 4-aminopiperidines as useful tools in the optimization of the 2-aminobenzo[a]quinolizine dipeptidyl peptidase IV inhibitors. Bioorg Med Chem Lett17:2966-70 (2007) [PubMed] Article
Lübbers, T; Böhringer, M; Gobbi, L; Hennig, M; Hunziker, D; Kuhn, B; Löffler, B; Mattei, P; Narquizian, R; Peters, JU; Ruff, Y; Wessel, HP; Wyss, P 1,3-disubstituted 4-aminopiperidines as useful tools in the optimization of the 2-aminobenzo[a]quinolizine dipeptidyl peptidase IV inhibitors. Bioorg Med Chem Lett17:2966-70 (2007) [PubMed] Article