| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Urokinase-type plasminogen activator |
|---|
| Ligand | BDBM50218291 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_448748 (CHEMBL897895) |
|---|
| Ki | >14000±n/a nM |
|---|
| Citation |  Qiao, JX; Wang, TC; Wang, GZ; Cheney, DL; He, K; Rendina, AR; Xin, B; Luettgen, JM; Knabb, RM; Wexler, RR; Lam, PY Enantiopure five-membered cyclicdiamine derivatives as potent and selective inhibitors of factor Xa. Improving in vitro metabolic stability via core modifications. Bioorg Med Chem Lett17:5041-8 (2007) [PubMed] Article Qiao, JX; Wang, TC; Wang, GZ; Cheney, DL; He, K; Rendina, AR; Xin, B; Luettgen, JM; Knabb, RM; Wexler, RR; Lam, PY Enantiopure five-membered cyclicdiamine derivatives as potent and selective inhibitors of factor Xa. Improving in vitro metabolic stability via core modifications. Bioorg Med Chem Lett17:5041-8 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Urokinase-type plasminogen activator |
|---|
| Name: | Urokinase-type plasminogen activator |
|---|
| Synonyms: | 3.4.21.73 | PLAU | U-plasminogen activator | UROK_HUMAN | Urokinase | Urokinase-type plasminogen activator (uPA) | Urokinase-type plasminogen activator chain B | Urokinase-type plasminogen activator long chain A | Urokinase-type plasminogen activator short chain A | Urokinase-type plasminogen activator/surface receptor | uPA |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48528.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00749 |
|---|
| Residue: | 431 |
|---|
| Sequence: | MRALLARLLLCVLVVSDSKGSNELHQVPSNCDCLNGGTCVSNKYFSNIHWCNCPKKFGGQ
HCEIDKSKTCYEGNGHFYRGKASTDTMGRPCLPWNSATVLQQTYHAHRSDALQLGLGKHN
YCRNPDNRRRPWCYVQVGLKLLVQECMVHDCADGKKPSSPPEELKFQCGQKTLRPRFKII
GGEFTTIENQPWFAAIYRRHRGGSVTYVCGGSLISPCWVISATHCFIDYPKKEDYIVYLG
RSRLNSNTQGEMKFEVENLILHKDYSADTLAHHNDIALLKIRSKEGRCAQPSRTIQTICL
PSMYNDPQFGTSCEITGFGKENSTDYLYPEQLKMTVVKLISHRECQQPHYYGSEVTTKML
CAADPQWKTDSCQGDSGGPLVCSLQGRMTLTGIVSWGRGCALKDKPGVYTRVSHFLPWIR
SHTKEENGLAL
|
|
|
|---|
| BDBM50218291 |
|---|
| n/a |
|---|
| Name | BDBM50218291 |
|---|
| Synonyms: | CHEMBL394129 | N-((3R,4S)-1-acetyl-4-(4-(2-oxopyridin-1(2H)-yl)benzamido)pyrrolidin-3-yl)-5-chlorothiophene-2-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H21ClN4O4S |
|---|
| Mol. Mass. | 484.955 |
|---|
| SMILES | CC(=O)N1C[C@@H](NC(=O)c2ccc(Cl)s2)[C@H](C1)NC(=O)c1ccc(cc1)-n1ccccc1=O |
|---|
| Structure | 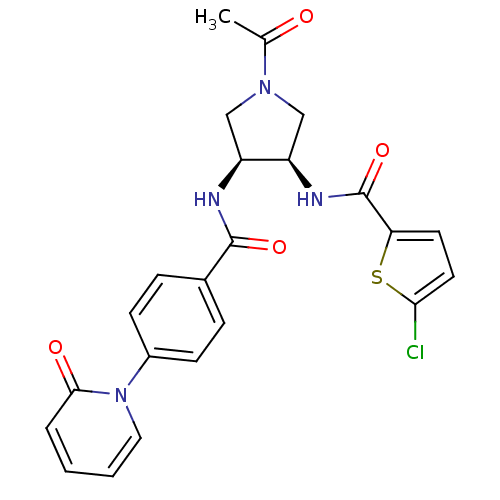
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Qiao, JX; Wang, TC; Wang, GZ; Cheney, DL; He, K; Rendina, AR; Xin, B; Luettgen, JM; Knabb, RM; Wexler, RR; Lam, PY Enantiopure five-membered cyclicdiamine derivatives as potent and selective inhibitors of factor Xa. Improving in vitro metabolic stability via core modifications. Bioorg Med Chem Lett17:5041-8 (2007) [PubMed] Article
Qiao, JX; Wang, TC; Wang, GZ; Cheney, DL; He, K; Rendina, AR; Xin, B; Luettgen, JM; Knabb, RM; Wexler, RR; Lam, PY Enantiopure five-membered cyclicdiamine derivatives as potent and selective inhibitors of factor Xa. Improving in vitro metabolic stability via core modifications. Bioorg Med Chem Lett17:5041-8 (2007) [PubMed] Article