

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | 5-hydroxytryptamine receptor 1A | ||
| Ligand | BDBM50252220 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_487560 (CHEMBL1013154) | ||
| Ki | 6.5±n/a nM | ||
| Citation |  Zhou, D; Zhou, P; Evrard, DA; Meagher, K; Webb, M; Harrison, BL; Huryn, DM; Golembieski, J; Hornby, GA; Schechter, LE; Smith, DL; Andree, TH; Mewshaw, RE Studies toward the discovery of the next generation of antidepressants. Part 6: Dual 5-HT1A receptor and serotonin transporter affinity within a class of arylpiperazinyl-cyclohexyl indole derivatives. Bioorg Med Chem16:6707-23 (2008) [PubMed] Article Zhou, D; Zhou, P; Evrard, DA; Meagher, K; Webb, M; Harrison, BL; Huryn, DM; Golembieski, J; Hornby, GA; Schechter, LE; Smith, DL; Andree, TH; Mewshaw, RE Studies toward the discovery of the next generation of antidepressants. Part 6: Dual 5-HT1A receptor and serotonin transporter affinity within a class of arylpiperazinyl-cyclohexyl indole derivatives. Bioorg Med Chem16:6707-23 (2008) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| 5-hydroxytryptamine receptor 1A | |||
| Name: | 5-hydroxytryptamine receptor 1A | ||
| Synonyms: | 5-HT-1A | 5-HT1A | 5-hydroxytryptamine receptor 1A (5-HT-1A) | 5HT1A_HUMAN | ADRB2RL1 | ADRBRL1 | Dopamine D2 receptor and serotonin 1a receptor | G-21 | HTR1A | Serotonin receptor 1A | ||
| Type: | n/a | ||
| Mol. Mass.: | 46122.49 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | n/a | ||
| Residue: | 422 | ||
| Sequence: |
| ||
| BDBM50252220 | |||
| n/a | |||
| Name | BDBM50252220 | ||
| Synonyms: | 8-{4-[(1,4-trans)-4-(5-Fluoro-1H-indol-3-yl)-cyclohexyl]-piperazin-1-yl}-quinoline | CHEMBL481753 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C27H29FN4 | ||
| Mol. Mass. | 428.5444 | ||
| SMILES | Fc1ccc2[nH]cc([C@H]3CC[C@@H](CC3)N3CCN(CC3)c3cccc4cccnc34)c2c1 |r,wU:8.7,wD:11.14,(-6.13,-34.96,;-5.82,-36.47,;-4.36,-36.96,;-4.06,-38.47,;-5.21,-39.48,;-5.23,-41.01,;-6.7,-41.47,;-7.59,-40.23,;-9.13,-40.21,;-9.91,-41.54,;-11.45,-41.52,;-12.21,-40.18,;-11.44,-38.85,;-9.89,-38.87,;-13.75,-40.17,;-14.53,-41.5,;-16.07,-41.48,;-16.82,-40.15,;-16.04,-38.82,;-14.51,-38.82,;-18.36,-40.14,;-19.13,-41.48,;-20.68,-41.47,;-21.45,-40.13,;-20.65,-38.8,;-21.42,-37.47,;-20.65,-36.15,;-19.11,-36.16,;-18.36,-37.49,;-19.12,-38.81,;-6.67,-38.98,;-6.97,-37.48,)| | ||
| Structure | 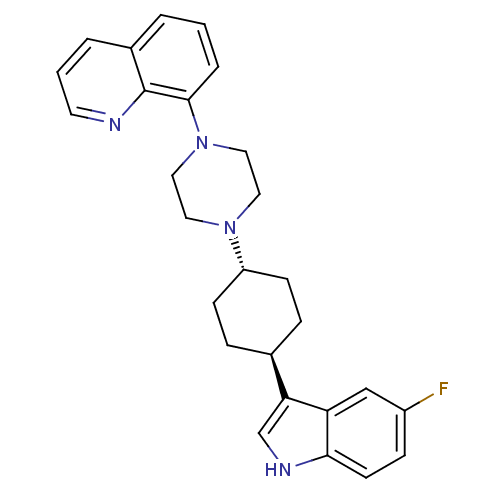
| ||