| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50300880 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_599659 (CHEMBL1042903) |
|---|
| Ki | 1520±n/a nM |
|---|
| Citation |  El-Tayeb, A; Iqbal, J; Behrenswerth, A; Romio, M; Schneider, M; Zimmermann, H; Schrader, J; Müller, CE Nucleoside-5'-monophosphates as prodrugs of adenosine A2A receptor agonists activated by ecto-5'-nucleotidase. J Med Chem52:7669-77 (2009) [PubMed] Article El-Tayeb, A; Iqbal, J; Behrenswerth, A; Romio, M; Schneider, M; Zimmermann, H; Schrader, J; Müller, CE Nucleoside-5'-monophosphates as prodrugs of adenosine A2A receptor agonists activated by ecto-5'-nucleotidase. J Med Chem52:7669-77 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | AA2AR_RAT | ADENOSINE A2a | Adenosine A2 receptor | Adenosine A2a receptor (A2a) | Adenosine Receptors A2a (A2a) | Adenosine receptor A2a and A3 | Adenosine receptors A2a | Adora2a | Rat striatal adenosine A2a receptor |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 45015.65 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat A2A receptors expressed in CHO cells. |
|---|
| Residue: | 410 |
|---|
| Sequence: | MGSSVYITVELAIAVLAILGNVLVCWAVWINSNLQNVTNFFVVSLAAADIAVGVLAIPFA
ITISTGFCAACHGCLFFACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGVRAKG
IIAICWVLSFAIGLTPMLGWNNCSQKDGNSTKTCGEGRVTCLFEDVVPMNYMVYYNFFAF
VLLPLLLMLAIYLRIFLAARRQLKQMESQPLPGERTRSTLQKEVHAAKSLAIIVGLFALC
WLPLHIINCFTFFCSTCRHAPPWLMYLAIILSHSNSVVNPFIYAYRIREFRQTFRKIIRT
HVLRRQEPFQAGGSSAWALAAHSTEGEQVSLRLNGHPLGVWANGSATHSGRRPNGYTLGL
GGGGSAQGSPRDVELPTQERQEGQEHPGLRGHLVQARVGASSWSSEFAPS
|
|
|
|---|
| BDBM50300880 |
|---|
| n/a |
|---|
| Name | BDBM50300880 |
|---|
| Synonyms: | (2R,3R,4S,5R)-2-(2-(allylthio)-6-amino-9H-purin-9-yl)-5-(hydroxymethyl)tetrahydrofuran-3,4-diol | CHEMBL578569 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H17N5O4S |
|---|
| Mol. Mass. | 339.37 |
|---|
| SMILES | Nc1nc(SCC=C)nc2n(cnc12)[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O |r| |
|---|
| Structure | 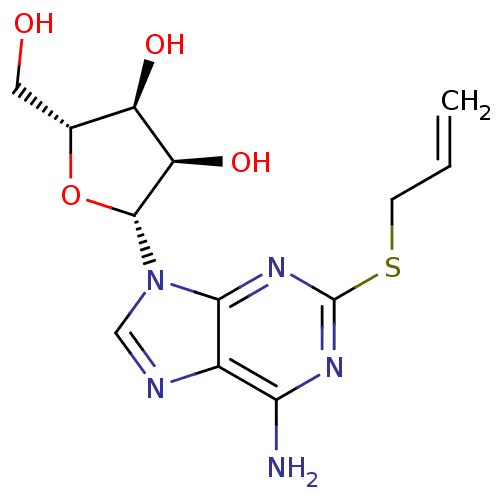
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 El-Tayeb, A; Iqbal, J; Behrenswerth, A; Romio, M; Schneider, M; Zimmermann, H; Schrader, J; Müller, CE Nucleoside-5'-monophosphates as prodrugs of adenosine A2A receptor agonists activated by ecto-5'-nucleotidase. J Med Chem52:7669-77 (2009) [PubMed] Article
El-Tayeb, A; Iqbal, J; Behrenswerth, A; Romio, M; Schneider, M; Zimmermann, H; Schrader, J; Müller, CE Nucleoside-5'-monophosphates as prodrugs of adenosine A2A receptor agonists activated by ecto-5'-nucleotidase. J Med Chem52:7669-77 (2009) [PubMed] Article