| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Urokinase-type plasminogen activator |
|---|
| Ligand | BDBM50307272 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_610622 (CHEMBL1069636) |
|---|
| Ki | 109±n/a nM |
|---|
| Citation |  Wang, S; Beck, R; Burd, A; Blench, T; Marlin, F; Ayele, T; Buxton, S; Dagostin, C; Malic, M; Joshi, R; Barry, J; Sajad, M; Cheung, C; Shaikh, S; Chahwala, S; Chander, C; Baumgartner, C; Holthoff, HP; Murray, E; Blackney, M; Giddings, A Structure based drug design: development of potent and selective factor IXa (FIXa) inhibitors. J Med Chem53:1473-82 (2010) [PubMed] Article Wang, S; Beck, R; Burd, A; Blench, T; Marlin, F; Ayele, T; Buxton, S; Dagostin, C; Malic, M; Joshi, R; Barry, J; Sajad, M; Cheung, C; Shaikh, S; Chahwala, S; Chander, C; Baumgartner, C; Holthoff, HP; Murray, E; Blackney, M; Giddings, A Structure based drug design: development of potent and selective factor IXa (FIXa) inhibitors. J Med Chem53:1473-82 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Urokinase-type plasminogen activator |
|---|
| Name: | Urokinase-type plasminogen activator |
|---|
| Synonyms: | 3.4.21.73 | PLAU | U-plasminogen activator | UROK_HUMAN | Urokinase | Urokinase-type plasminogen activator (uPA) | Urokinase-type plasminogen activator chain B | Urokinase-type plasminogen activator long chain A | Urokinase-type plasminogen activator short chain A | Urokinase-type plasminogen activator/surface receptor | uPA |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48528.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00749 |
|---|
| Residue: | 431 |
|---|
| Sequence: | MRALLARLLLCVLVVSDSKGSNELHQVPSNCDCLNGGTCVSNKYFSNIHWCNCPKKFGGQ
HCEIDKSKTCYEGNGHFYRGKASTDTMGRPCLPWNSATVLQQTYHAHRSDALQLGLGKHN
YCRNPDNRRRPWCYVQVGLKLLVQECMVHDCADGKKPSSPPEELKFQCGQKTLRPRFKII
GGEFTTIENQPWFAAIYRRHRGGSVTYVCGGSLISPCWVISATHCFIDYPKKEDYIVYLG
RSRLNSNTQGEMKFEVENLILHKDYSADTLAHHNDIALLKIRSKEGRCAQPSRTIQTICL
PSMYNDPQFGTSCEITGFGKENSTDYLYPEQLKMTVVKLISHRECQQPHYYGSEVTTKML
CAADPQWKTDSCQGDSGGPLVCSLQGRMTLTGIVSWGRGCALKDKPGVYTRVSHFLPWIR
SHTKEENGLAL
|
|
|
|---|
| BDBM50307272 |
|---|
| n/a |
|---|
| Name | BDBM50307272 |
|---|
| Synonyms: | (5-Methoxymethyl-2-methyl-2H-pyrazol-3-yl)-carbamic acid 2-(2-carbamimidoylbenzo[b]thiophen-4-yloxy)-2-phenyl-ethyl ester | CHEMBL601870 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H25N5O4S |
|---|
| Mol. Mass. | 479.551 |
|---|
| SMILES | COCc1cc(NC(=O)OCC(Oc2cccc3sc(cc23)C(N)=N)c2ccccc2)n(C)n1 |
|---|
| Structure | 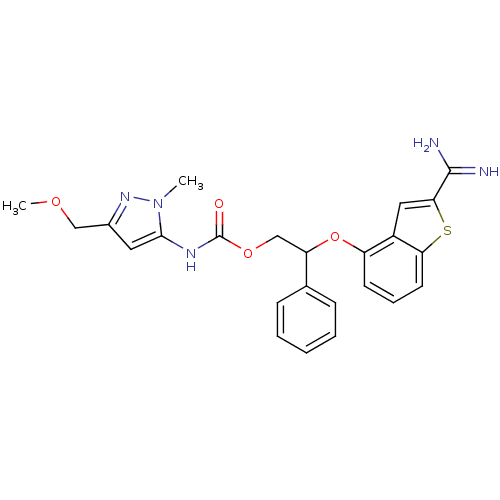
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wang, S; Beck, R; Burd, A; Blench, T; Marlin, F; Ayele, T; Buxton, S; Dagostin, C; Malic, M; Joshi, R; Barry, J; Sajad, M; Cheung, C; Shaikh, S; Chahwala, S; Chander, C; Baumgartner, C; Holthoff, HP; Murray, E; Blackney, M; Giddings, A Structure based drug design: development of potent and selective factor IXa (FIXa) inhibitors. J Med Chem53:1473-82 (2010) [PubMed] Article
Wang, S; Beck, R; Burd, A; Blench, T; Marlin, F; Ayele, T; Buxton, S; Dagostin, C; Malic, M; Joshi, R; Barry, J; Sajad, M; Cheung, C; Shaikh, S; Chahwala, S; Chander, C; Baumgartner, C; Holthoff, HP; Murray, E; Blackney, M; Giddings, A Structure based drug design: development of potent and selective factor IXa (FIXa) inhibitors. J Med Chem53:1473-82 (2010) [PubMed] Article