| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM50324758 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_651359 (CHEMBL1227179) |
|---|
| Ki | 10±n/a nM |
|---|
| Citation |  Anselm, L; Banner, DW; Benz, J; Zbinden, KG; Himber, J; Hilpert, H; Huber, W; Kuhn, B; Mary, JL; Otteneder, MB; Panday, N; Ricklin, F; Stahl, M; Thomi, S; Haap, W Discovery of a factor Xa inhibitor (3R,4R)-1-(2,2-difluoro-ethyl)-pyrrolidine-3,4-dicarboxylic acid 3-[(5-chloro-pyridin-2-yl)-amide] 4-[[2-fluoro-4-(2-oxo-2H-pyridin-1-yl)-phenyl]-amide] as a clinical candidate. Bioorg Med Chem Lett20:5313-9 (2010) [PubMed] Article Anselm, L; Banner, DW; Benz, J; Zbinden, KG; Himber, J; Hilpert, H; Huber, W; Kuhn, B; Mary, JL; Otteneder, MB; Panday, N; Ricklin, F; Stahl, M; Thomi, S; Haap, W Discovery of a factor Xa inhibitor (3R,4R)-1-(2,2-difluoro-ethyl)-pyrrolidine-3,4-dicarboxylic acid 3-[(5-chloro-pyridin-2-yl)-amide] 4-[[2-fluoro-4-(2-oxo-2H-pyridin-1-yl)-phenyl]-amide] as a clinical candidate. Bioorg Med Chem Lett20:5313-9 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM50324758 |
|---|
| n/a |
|---|
| Name | BDBM50324758 |
|---|
| Synonyms: | (3R,4R)-N3-(4-chlorophenyl)-N4-(2-fluoro-4-(2-oxopyridin-1(2H)-yl)phenyl)-1-(isopropylsulfonyl)pyrrolidine-3,4-dicarboxamide | CHEMBL1221674 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H26ClFN4O5S |
|---|
| Mol. Mass. | 561.025 |
|---|
| SMILES | CC(C)S(=O)(=O)N1C[C@@H]([C@H](C1)C(=O)Nc1ccc(cc1F)-n1ccccc1=O)C(=O)Nc1ccc(Cl)cc1 |r| |
|---|
| Structure | 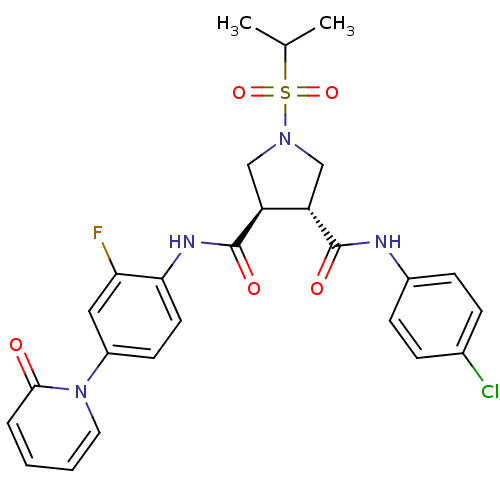
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Anselm, L; Banner, DW; Benz, J; Zbinden, KG; Himber, J; Hilpert, H; Huber, W; Kuhn, B; Mary, JL; Otteneder, MB; Panday, N; Ricklin, F; Stahl, M; Thomi, S; Haap, W Discovery of a factor Xa inhibitor (3R,4R)-1-(2,2-difluoro-ethyl)-pyrrolidine-3,4-dicarboxylic acid 3-[(5-chloro-pyridin-2-yl)-amide] 4-[[2-fluoro-4-(2-oxo-2H-pyridin-1-yl)-phenyl]-amide] as a clinical candidate. Bioorg Med Chem Lett20:5313-9 (2010) [PubMed] Article
Anselm, L; Banner, DW; Benz, J; Zbinden, KG; Himber, J; Hilpert, H; Huber, W; Kuhn, B; Mary, JL; Otteneder, MB; Panday, N; Ricklin, F; Stahl, M; Thomi, S; Haap, W Discovery of a factor Xa inhibitor (3R,4R)-1-(2,2-difluoro-ethyl)-pyrrolidine-3,4-dicarboxylic acid 3-[(5-chloro-pyridin-2-yl)-amide] 4-[[2-fluoro-4-(2-oxo-2H-pyridin-1-yl)-phenyl]-amide] as a clinical candidate. Bioorg Med Chem Lett20:5313-9 (2010) [PubMed] Article