| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50326292 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_659957 (CHEMBL1247358) |
|---|
| IC50 | 6500±n/a nM |
|---|
| Citation |  Ishikawa, M; Watanabe, T; Kudo, T; Yokoyama, F; Yamauchi, M; Kato, K; Kakui, N; Sato, Y Investigation of the histamine H3 receptor binding site. Design and synthesis of hybrid agonists with a lipophilic side chain. J Med Chem53:6445-56 (2010) [PubMed] Article Ishikawa, M; Watanabe, T; Kudo, T; Yokoyama, F; Yamauchi, M; Kato, K; Kakui, N; Sato, Y Investigation of the histamine H3 receptor binding site. Design and synthesis of hybrid agonists with a lipophilic side chain. J Med Chem53:6445-56 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50326292 |
|---|
| n/a |
|---|
| Name | BDBM50326292 |
|---|
| Synonyms: | 4-(2-(4-tert-Butylphenylthio)-1-(1H-imidazol-4-yl)ethyl)piperidine | CHEMBL1243333 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H29N3S |
|---|
| Mol. Mass. | 343.529 |
|---|
| SMILES | CC(C)(C)c1ccc(SCC(C2CCNCC2)c2cnc[nH]2)cc1 |
|---|
| Structure | 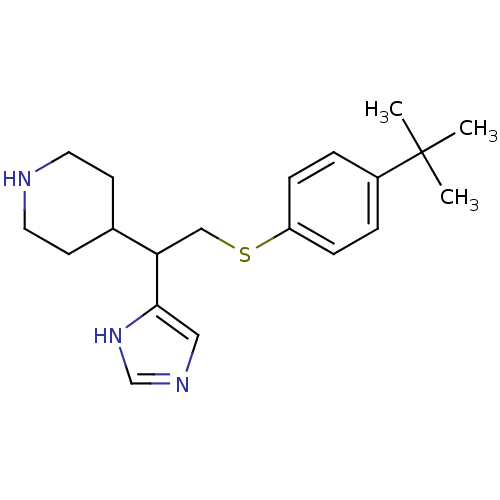
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ishikawa, M; Watanabe, T; Kudo, T; Yokoyama, F; Yamauchi, M; Kato, K; Kakui, N; Sato, Y Investigation of the histamine H3 receptor binding site. Design and synthesis of hybrid agonists with a lipophilic side chain. J Med Chem53:6445-56 (2010) [PubMed] Article
Ishikawa, M; Watanabe, T; Kudo, T; Yokoyama, F; Yamauchi, M; Kato, K; Kakui, N; Sato, Y Investigation of the histamine H3 receptor binding site. Design and synthesis of hybrid agonists with a lipophilic side chain. J Med Chem53:6445-56 (2010) [PubMed] Article