| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Nicotinamide phosphoribosyltransferase |
|---|
| Ligand | BDBM50117198 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_754944 (CHEMBL1805766) |
|---|
| IC50 | 0.31±n/a nM |
|---|
| Citation |  Lockman, JW; Murphy, BR; Zigar, DF; Judd, WR; Slattum, PM; Gao, ZH; Ostanin, K; Green, J; McKinnon, R; Terry-Lorenzo, RT; Fleischer, TC; Boniface, JJ; Shenderovich, M; Willardsen, JA Analogues of 4-[(7-Bromo-2-methyl-4-oxo-3H-quinazolin-6-yl)methylprop-2-ynylamino]-N-(3-pyridylmethyl)benzamide (CB-30865) as potent inhibitors of nicotinamide phosphoribosyltransferase (Nampt). J Med Chem53:8734-46 (2010) [PubMed] Article Lockman, JW; Murphy, BR; Zigar, DF; Judd, WR; Slattum, PM; Gao, ZH; Ostanin, K; Green, J; McKinnon, R; Terry-Lorenzo, RT; Fleischer, TC; Boniface, JJ; Shenderovich, M; Willardsen, JA Analogues of 4-[(7-Bromo-2-methyl-4-oxo-3H-quinazolin-6-yl)methylprop-2-ynylamino]-N-(3-pyridylmethyl)benzamide (CB-30865) as potent inhibitors of nicotinamide phosphoribosyltransferase (Nampt). J Med Chem53:8734-46 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Nicotinamide phosphoribosyltransferase |
|---|
| Name: | Nicotinamide phosphoribosyltransferase |
|---|
| Synonyms: | NAMPT | NAMPT_HUMAN | NAmPRTase | Nicotinamide phosphoribosyltransferase | Nicotinamide phosphoribosyltransferase (NAMPT) | PBEF | PBEF1 | Pre-B-cell colony-enhancing factor 1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55524.98 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P43490 |
|---|
| Residue: | 491 |
|---|
| Sequence: | MNPAAEAEFNILLATDSYKVTHYKQYPPNTSKVYSYFECREKKTENSKLRKVKYEETVFY
GLQYILNKYLKGKVVTKEKIQEAKDVYKEHFQDDVFNEKGWNYILEKYDGHLPIEIKAVP
EGFVIPRGNVLFTVENTDPECYWLTNWIETILVQSWYPITVATNSREQKKILAKYLLETS
GNLDGLEYKLHDFGYRGVSSQETAGIGASAHLVNFKGTDTVAGLALIKKYYGTKDPVPGY
SVPAAEHSTITAWGKDHEKDAFEHIVTQFSSVPVSVVSDSYDIYNACEKIWGEDLRHLIV
SRSTQAPLIIRPDSGNPLDTVLKVLEILGKKFPVTENSKGYKLLPPYLRVIQGDGVDINT
LQEIVEGMKQKMWSIENIAFGSGGGLLQKLTRDLLNCSFKCSYVVTNGLGINVFKDPVAD
PNKRSKKGRLSLHRTPAGNFVTLEEGKGDLEEYGQDLLHTVFKNGKVTKSYSFDEIRKNA
QLNIELEAAHH
|
|
|
|---|
| BDBM50117198 |
|---|
| n/a |
|---|
| Name | BDBM50117198 |
|---|
| Synonyms: | 4-{[7-Chloro-3-methyl-2-(4-methyl-piperazin-1-ylmethyl)-4-oxo-3,4-dihydro-quinazolin-6-ylmethyl]-prop-2-ynyl-amino}-N-pyridin-3-ylmethyl-benzamide | CHEMBL123756 | MPI-0479626 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H34ClN7O2 |
|---|
| Mol. Mass. | 584.111 |
|---|
| SMILES | CN1CCN(Cc2nc3cc(Cl)c(CN(CC#C)c4ccc(cc4)C(=O)NCc4cccnc4)cc3c(=O)n2C)CC1 |
|---|
| Structure | 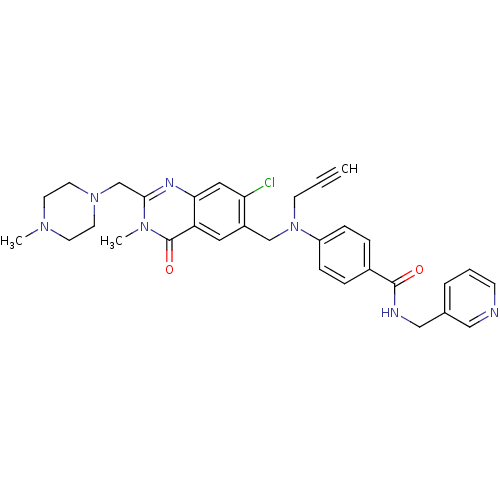
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lockman, JW; Murphy, BR; Zigar, DF; Judd, WR; Slattum, PM; Gao, ZH; Ostanin, K; Green, J; McKinnon, R; Terry-Lorenzo, RT; Fleischer, TC; Boniface, JJ; Shenderovich, M; Willardsen, JA Analogues of 4-[(7-Bromo-2-methyl-4-oxo-3H-quinazolin-6-yl)methylprop-2-ynylamino]-N-(3-pyridylmethyl)benzamide (CB-30865) as potent inhibitors of nicotinamide phosphoribosyltransferase (Nampt). J Med Chem53:8734-46 (2010) [PubMed] Article
Lockman, JW; Murphy, BR; Zigar, DF; Judd, WR; Slattum, PM; Gao, ZH; Ostanin, K; Green, J; McKinnon, R; Terry-Lorenzo, RT; Fleischer, TC; Boniface, JJ; Shenderovich, M; Willardsen, JA Analogues of 4-[(7-Bromo-2-methyl-4-oxo-3H-quinazolin-6-yl)methylprop-2-ynylamino]-N-(3-pyridylmethyl)benzamide (CB-30865) as potent inhibitors of nicotinamide phosphoribosyltransferase (Nampt). J Med Chem53:8734-46 (2010) [PubMed] Article