| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Progesterone receptor |
|---|
| Ligand | BDBM50086545 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_159559 (CHEMBL765847) |
|---|
| Ki | 237±n/a nM |
|---|
| Citation |  Zhi, L; Tegley, CM; Pio, B; West, SJ; Marschke, KB; Mais, DE; Jones, TK Nonsteroidal progesterone receptor antagonists based on 6-thiophenehydroquinolines. Bioorg Med Chem Lett10:415-8 (2000) [PubMed] Zhi, L; Tegley, CM; Pio, B; West, SJ; Marschke, KB; Mais, DE; Jones, TK Nonsteroidal progesterone receptor antagonists based on 6-thiophenehydroquinolines. Bioorg Med Chem Lett10:415-8 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Progesterone receptor |
|---|
| Name: | Progesterone receptor |
|---|
| Synonyms: | NR3C3 | Nuclear receptor subfamily 3 group C member 3 | PGR | PR | PRGR_HUMAN | progesterone |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 98979.52 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06401 |
|---|
| Residue: | 933 |
|---|
| Sequence: | MTELKAKGPRAPHVAGGPPSPEVGSPLLCRPAAGPFPGSQTSDTLPEVSAIPISLDGLLF
PRPCQGQDPSDEKTQDQQSLSDVEGAYSRAEATRGAGGSSSSPPEKDSGLLDSVLDTLLA
PSGPGQSQPSPPACEVTSSWCLFGPELPEDPPAAPATQRVLSPLMSRSGCKVGDSSGTAA
AHKVLPRGLSPARQLLLPASESPHWSGAPVKPSPQAAAVEVEEEDGSESEESAGPLLKGK
PRALGGAAAGGGAAAVPPGAAAGGVALVPKEDSRFSAPRVALVEQDAPMAPGRSPLATTV
MDFIHVPILPLNHALLAARTRQLLEDESYDGGAGAASAFAPPRSSPCASSTPVAVGDFPD
CAYPPDAEPKDDAYPLYSDFQPPALKIKEEEEGAEASARSPRSYLVAGANPAAFPDFPLG
PPPPLPPRATPSRPGEAAVTAAPASASVSSASSSGSTLECILYKAEGAPPQQGPFAPPPC
KAPGASGCLLPRDGLPSTSASAAAAGAAPALYPALGLNGLPQLGYQAAVLKEGLPQVYPP
YLNYLRPDSEASQSPQYSFESLPQKICLICGDEASGCHYGVLTCGSCKVFFKRAMEGQHN
YLCAGRNDCIVDKIRRKNCPACRLRKCCQAGMVLGGRKFKKFNKVRVVRALDAVALPQPV
GVPNESQALSQRFTFSPGQDIQLIPPLINLLMSIEPDVIYAGHDNTKPDTSSSLLTSLNQ
LGERQLLSVVKWSKSLPGFRNLHIDDQITLIQYSWMSLMVFGLGWRSYKHVSGQMLYFAP
DLILNEQRMKESSFYSLCLTMWQIPQEFVKLQVSQEEFLCMKVLLLLNTIPLEGLRSQTQ
FEEMRSSYIRELIKAIGLRQKGVVSSSQRFYQLTKLLDNLHDLVKQLHLYCLNTFIQSRA
LSVEFPEMMSEVIAAQLPKILAGMVKPLLFHKK
|
|
|
|---|
| BDBM50086545 |
|---|
| n/a |
|---|
| Name | BDBM50086545 |
|---|
| Synonyms: | 2,2,4-Trimethyl-6-thiophen-2-yl-1,2-dihydro-quinoline | 2,2,4-trimethyl-6-(thiophen-2-yl)-1,2-dihydroquinoline | CHEMBL337199 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H17NS |
|---|
| Mol. Mass. | 255.378 |
|---|
| SMILES | CC1=CC(C)(C)Nc2ccc(cc12)-c1cccs1 |t:1| |
|---|
| Structure | 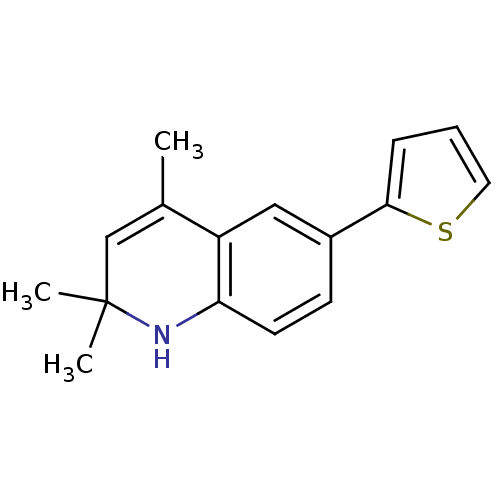
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhi, L; Tegley, CM; Pio, B; West, SJ; Marschke, KB; Mais, DE; Jones, TK Nonsteroidal progesterone receptor antagonists based on 6-thiophenehydroquinolines. Bioorg Med Chem Lett10:415-8 (2000) [PubMed]
Zhi, L; Tegley, CM; Pio, B; West, SJ; Marschke, KB; Mais, DE; Jones, TK Nonsteroidal progesterone receptor antagonists based on 6-thiophenehydroquinolines. Bioorg Med Chem Lett10:415-8 (2000) [PubMed]