| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1B |
|---|
| Ligand | BDBM50145332 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_1343 |
|---|
| Ki | 316±n/a nM |
|---|
| Citation |  Timms, GH; Boot, JR; Broadmore, RJ; Carney, SL; Cooper, J; Findlay, JD; Gilmore, J; Mitchell, S; Moore, NA; Pullar, I; Sanger, GJ; Tomlinson, R; Tree, BB; Wedley, S SAR development of a selective 5-HT1D antagonist/serotonin reuptake inhibitor lead using rapid parallel synthesis. Bioorg Med Chem Lett14:2469-72 (2004) [PubMed] Article Timms, GH; Boot, JR; Broadmore, RJ; Carney, SL; Cooper, J; Findlay, JD; Gilmore, J; Mitchell, S; Moore, NA; Pullar, I; Sanger, GJ; Tomlinson, R; Tree, BB; Wedley, S SAR development of a selective 5-HT1D antagonist/serotonin reuptake inhibitor lead using rapid parallel synthesis. Bioorg Med Chem Lett14:2469-72 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1B |
|---|
| Name: | 5-hydroxytryptamine receptor 1B |
|---|
| Synonyms: | 5-HT-1B | 5-HT-1D-beta | 5-HT1B | 5-hydroxytryptamine receptor 1B (5-HT1B) | 5HT1B_HUMAN | HTR1B | HTR1DB | S12 | Serotonin (5-HT) receptor | Serotonin 1D beta receptor | Serotonin Receptor 1B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 43579.17 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells |
|---|
| Residue: | 390 |
|---|
| Sequence: | MEEPGAQCAPPPPAGSETWVPQANLSSAPSQNCSAKDYIYQDSISLPWKVLLVMLLALIT
LATTLSNAFVIATVYRTRKLHTPANYLIASLAVTDLLVSILVMPISTMYTVTGRWTLGQV
VCDFWLSSDITCCTASILHLCVIALDRYWAITDAVEYSAKRTPKRAAVMIALVWVFSISI
SLPPFFWRQAKAEEEVSECVVNTDHILYTVYSTVGAFYFPTLLLIALYGRIYVEARSRIL
KQTPNRTGKRLTRAQLITDSPGSTSSVTSINSRVPDVPSESGSPVYVNQVKVRVSDALLE
KKKLMAARERKATKTLGIILGAFIVCWLPFFIISLVMPICKDACWFHLAIFDFFTWLGYL
NSLINPIIYTMSNEDFKQAFHKLIRFKCTS
|
|
|
|---|
| BDBM50145332 |
|---|
| n/a |
|---|
| Name | BDBM50145332 |
|---|
| Synonyms: | 1-{2-[4-(5-Methoxy-1H-indol-3-yl)-3,6-dihydro-2H-pyridin-1-yl]-ethyl}-isochroman-6-carboxylic acid amide | CHEMBL84171 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H29N3O3 |
|---|
| Mol. Mass. | 431.5268 |
|---|
| SMILES | COc1ccc2[nH]cc(C3=CCN(CCC4OCCc5cc(ccc45)C(N)=O)CC3)c2c1 |t:9| |
|---|
| Structure | 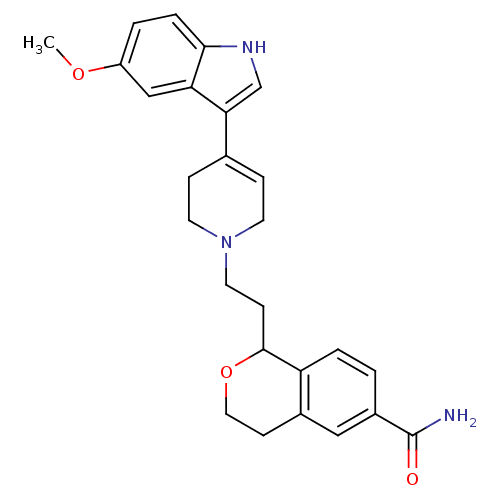
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Timms, GH; Boot, JR; Broadmore, RJ; Carney, SL; Cooper, J; Findlay, JD; Gilmore, J; Mitchell, S; Moore, NA; Pullar, I; Sanger, GJ; Tomlinson, R; Tree, BB; Wedley, S SAR development of a selective 5-HT1D antagonist/serotonin reuptake inhibitor lead using rapid parallel synthesis. Bioorg Med Chem Lett14:2469-72 (2004) [PubMed] Article
Timms, GH; Boot, JR; Broadmore, RJ; Carney, SL; Cooper, J; Findlay, JD; Gilmore, J; Mitchell, S; Moore, NA; Pullar, I; Sanger, GJ; Tomlinson, R; Tree, BB; Wedley, S SAR development of a selective 5-HT1D antagonist/serotonin reuptake inhibitor lead using rapid parallel synthesis. Bioorg Med Chem Lett14:2469-72 (2004) [PubMed] Article