

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cholecystokinin receptor type A | ||
| Ligand | BDBM50005815 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEBML_50060 | ||
| IC50 | 1230±n/a nM | ||
| Citation |  Fincham, CI; Higginbottom, M; Hill, DR; Horwell, DC; O'Toole, JC; Ratcliffe, GS; Rees, DC; Roberts, E Amide bond replacements incorporated into CCK-B selective"dipeptoids". J Med Chem35:1472-84 (1992) [PubMed] Fincham, CI; Higginbottom, M; Hill, DR; Horwell, DC; O'Toole, JC; Ratcliffe, GS; Rees, DC; Roberts, E Amide bond replacements incorporated into CCK-B selective"dipeptoids". J Med Chem35:1472-84 (1992) [PubMed] | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cholecystokinin receptor type A | |||
| Name: | Cholecystokinin receptor type A | ||
| Synonyms: | CCKAR_RAT | Cckar | Cholecystokinin peripheral | Cholecystokinin receptor | Cholecystokinin receptor type A | ||
| Type: | Enzyme Catalytic Domain | ||
| Mol. Mass.: | 49676.37 | ||
| Organism: | RAT | ||
| Description: | Cholecystokinin central 0 RAT::P30551 | ||
| Residue: | 444 | ||
| Sequence: |
| ||
| BDBM50005815 | |||
| n/a | |||
| Name | BDBM50005815 | ||
| Synonyms: | CHEMBL280502 | [1-(2-Hydroxymethyl-3-phenyl-propionylamino)-2-(1H-indol-3-yl)-ethyl]-carbamic acid adamantan-2-yl ester;0.25H2O | [1-(2-Hydroxymethyl-3-phenyl-propionylamino)-2-(1H-indol-3-yl)-ethyl]-carbamic acid adamantan-2-yl ester;0.2methyl tetrachloride | ||
| Type | Small organic molecule | ||
| Emp. Form. | C31H37N3O4 | ||
| Mol. Mass. | 515.6432 | ||
| SMILES | OCC(Cc1ccccc1)C(=O)NC(Cc1c[nH]c2ccccc12)NC(=O)OC1[C@H]2C[C@@H]3C[C@@H](C[C@H]1C3)C2 |wU:31.33,35.37,wD:33.41,29.32,TLB:27:28:34:31.36.32,THB:30:29:34:31.36.32,30:31:34:28.29.37,(18.97,-6.87,;17.43,-6.97,;16.76,-8.35,;17.62,-9.63,;19.16,-9.51,;20.02,-10.79,;21.54,-10.68,;22.21,-9.28,;21.34,-8.02,;19.8,-8.13,;15.22,-8.47,;14.54,-9.86,;14.35,-7.2,;12.84,-7.31,;11.59,-6.42,;11.75,-4.88,;10.6,-3.86,;11.21,-2.45,;12.75,-2.6,;13.87,-1.55,;15.35,-2.02,;15.67,-3.54,;14.53,-4.57,;13.07,-4.11,;12.06,-8.63,;10.52,-8.61,;9.76,-7.26,;9.73,-9.92,;8.19,-9.89,;6.81,-9.31,;5.46,-9.8,;5.46,-11.27,;4.28,-12.55,;5.77,-12.14,;7.16,-12.7,;8.19,-11.43,;6.78,-11.77,;5.75,-10.55,)| | ||
| Structure | 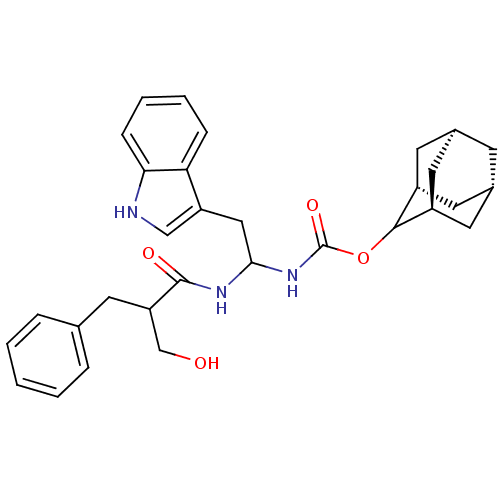
| ||