| Reaction Details |
|---|
 | Report a problem with these data |
| Target | D(1A) dopamine receptor |
|---|
| Ligand | BDBM50026957 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_60334 (CHEMBL673545) |
|---|
| Ki | 18±n/a nM |
|---|
| Citation |  el Ahmad, Y; Laurent, E; Maillet, P; Talab, A; Teste, JF; Dokhan, R; Tran, G; Ollivier, R New benzocycloalkylpiperazines, potent and selective 5-HT1A receptor ligands. J Med Chem40:952-60 (1997) [PubMed] Article el Ahmad, Y; Laurent, E; Maillet, P; Talab, A; Teste, JF; Dokhan, R; Tran, G; Ollivier, R New benzocycloalkylpiperazines, potent and selective 5-HT1A receptor ligands. J Med Chem40:952-60 (1997) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| D(1A) dopamine receptor |
|---|
| Name: | D(1A) dopamine receptor |
|---|
| Synonyms: | D(1A) dopamine receptor | D1AR | DOPAMINE D1 | DRD1 | DRD1_BOVIN | Dopamine receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 49330.65 |
|---|
| Organism: | BOVINE |
|---|
| Description: | DOPAMINE D1 DRD1 BOVINE::Q95136 |
|---|
| Residue: | 446 |
|---|
| Sequence: | MRTLNTSTMEGTGLVAERDFSFRILTACFLSLLILSTLLGNTLVCAAVIRFRHLRSKVTN
FFVISLAVSDLLVAVLVMPWKAVAEIAGFWPFGSFCNIWVAFDIMCSTASILNLCVISVD
RYWAISSPFRYERKMTPKAAFILISVAWTLSVLISFIPVQLSWHKAKPTGPSEGNATSLG
KTINNCDSSLSRTYAISSSLISFYIPVAIMIVTYTRIYRIAQKQIRRISALERAAVHAKN
CQTTTGNGNPMECSQPESSFKMSFKRETKVLKTLSVIMGVFVCCWLPFFILNCMVPFCGS
GETKPFCIDSITFDVFVWFGWANSSLNPIIYAFNADFRKAFSTLLGCYRLCPTTNNAIET
VSINNNGAVVFSSHHEPRGSISKDCNVVYLIPHAVGSSEGLKKEEAVGIAKPLEKLSPAL
SVILDYDTDVSLEKIQPITQNGQHPT
|
|
|
|---|
| BDBM50026957 |
|---|
| n/a |
|---|
| Name | BDBM50026957 |
|---|
| Synonyms: | (cis) 2-{4-[3-(2-Trifluoromethyl-thioxanthen-9-ylidene)-propyl]-piperazin-1-yl}-ethanol(Flupentixol) | (trans-flupenthixol)2-{4-[3-(2-Trifluoromethyl-thioxanthen-9-ylidene)-propyl]-piperazin-1-yl}-ethanol | 2-(4-{3-[2-Trifluoromethyl-thioxanthen-(9E)-ylidene]-propyl}-piperazin-1-yl)-ethanol | 2-{4-[3-(2-Trifluoromethyl-thioxanthen-9-ylidene)-propyl]-piperazin-1-yl}-ethanol | FLUPENTHIXOL, Alpha | FLUPENTHIXOL2-{4-[3-(2-Trifluoromethyl-thioxanthen-9-ylidene)-propyl]-piperazin-1-yl}-ethanol | Flupenthixol | Flupenthixol, Beta |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H25F3N2OS |
|---|
| Mol. Mass. | 434.518 |
|---|
| SMILES | OCCN1CCN(CC\C=C2/c3ccccc3Sc3ccc(cc23)C(F)(F)F)CC1 |
|---|
| Structure | 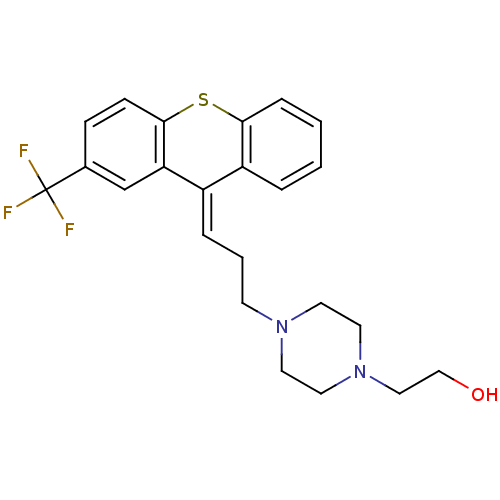
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 el Ahmad, Y; Laurent, E; Maillet, P; Talab, A; Teste, JF; Dokhan, R; Tran, G; Ollivier, R New benzocycloalkylpiperazines, potent and selective 5-HT1A receptor ligands. J Med Chem40:952-60 (1997) [PubMed] Article
el Ahmad, Y; Laurent, E; Maillet, P; Talab, A; Teste, JF; Dokhan, R; Tran, G; Ollivier, R New benzocycloalkylpiperazines, potent and selective 5-HT1A receptor ligands. J Med Chem40:952-60 (1997) [PubMed] Article