| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM50157332 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_304697 (CHEMBL827049) |
|---|
| IC50 | 190±n/a nM |
|---|
| Citation |  Ueno, H; Yokota, K; Hoshi, J; Yasue, K; Hayashi, M; Hase, Y; Uchida, I; Aisaka, K; Katoh, S; Cho, H Synthesis and structure-activity relationships of novel selective factor Xa inhibitors with a tetrahydroisoquinoline ring. J Med Chem48:3586-604 (2005) [PubMed] Article Ueno, H; Yokota, K; Hoshi, J; Yasue, K; Hayashi, M; Hase, Y; Uchida, I; Aisaka, K; Katoh, S; Cho, H Synthesis and structure-activity relationships of novel selective factor Xa inhibitors with a tetrahydroisoquinoline ring. J Med Chem48:3586-604 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM50157332 |
|---|
| n/a |
|---|
| Name | BDBM50157332 |
|---|
| Synonyms: | 3-[4-(2-Carbamimidoyl-1,2,3,4-tetrahydro-isoquinolin-7-yloxymethyl)-3,4,5,6-tetrahydro-2H-[1,4'']bipyridinyl-3''-yl]-propionic acid ethyl ester; dihydrochloride | 3-[4-(2-Carbamimidoyl-1,2,3,4-tetrahydro-isoquinolin-7-yloxymethyl)-3,4,5,6-tetrahydro-2H-[1,4']bipyridinyl-3'-yl]-propionic acid ethyl ester; dihydrochloride | CHEMBL540102 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H35N5O3 |
|---|
| Mol. Mass. | 465.5878 |
|---|
| SMILES | CCOC(=O)CCc1cnccc1N1CCC(COc2ccc3CCN(Cc3c2)C(N)=N)CC1 |
|---|
| Structure | 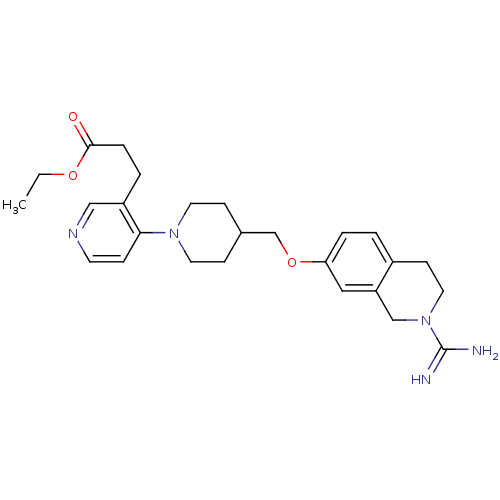
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ueno, H; Yokota, K; Hoshi, J; Yasue, K; Hayashi, M; Hase, Y; Uchida, I; Aisaka, K; Katoh, S; Cho, H Synthesis and structure-activity relationships of novel selective factor Xa inhibitors with a tetrahydroisoquinoline ring. J Med Chem48:3586-604 (2005) [PubMed] Article
Ueno, H; Yokota, K; Hoshi, J; Yasue, K; Hayashi, M; Hase, Y; Uchida, I; Aisaka, K; Katoh, S; Cho, H Synthesis and structure-activity relationships of novel selective factor Xa inhibitors with a tetrahydroisoquinoline ring. J Med Chem48:3586-604 (2005) [PubMed] Article