| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glutamate receptor ionotropic, kainate 2 |
|---|
| Ligand | BDBM50373179 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_467211 (CHEMBL928082) |
|---|
| EC50 | >1000000±n/a nM |
|---|
| Citation |  Pinto, A; Conti, P; De Amici, M; Tamborini, L; Madsen, U; Nielsen, B; Christesen, T; Bräuner-Osborne, H; De Micheli, C Synthesis and pharmacological characterization at glutamate receptors of the four enantiopure isomers of tricholomic acid. J Med Chem51:2311-5 (2008) [PubMed] Article Pinto, A; Conti, P; De Amici, M; Tamborini, L; Madsen, U; Nielsen, B; Christesen, T; Bräuner-Osborne, H; De Micheli, C Synthesis and pharmacological characterization at glutamate receptors of the four enantiopure isomers of tricholomic acid. J Med Chem51:2311-5 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glutamate receptor ionotropic, kainate 2 |
|---|
| Name: | Glutamate receptor ionotropic, kainate 2 |
|---|
| Synonyms: | GRIK2_RAT | Glur6 | Glutamate receptor ionotropic kainate 2 | Grik2 | Ionotropic glutamate receptor kainate 2/5 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 102478.61 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_936439 |
|---|
| Residue: | 908 |
|---|
| Sequence: | MKIISPVLSNLVFSRSIKVLLCLLWIGYSQGTTHVLRFGGIFEYVESGPMGAEELAFRFA
VNTINRNRTLLPNTTLTYDTQKINLYDSFEASKKACDQLSLGVAAIFGPSHSSSANAVQS
ICNALGVPHIQTRWKHQVSDNKDSFYVSLYPDFSSLSRAILDLVQFFKWKTVTVVYDDST
GLIRLQELIKAPSRYNLRLKIRQLPADTKDAKPLLKEMKRGKEFHVIFDCSHEMAAGILK
QALAMGMMTEYYHYIFTTLDLFALDVEPYRYSGVNMTGFRILNTENTQVSSIIEKWSMER
LQAPPKPDSGLLDGFMTTDAALMYDAVHVVSVAVQQFPQMTVSSLQCNRHKPWRFGTRFM
SLIKEAHWEGLTGRITFNKTNGLRTDFDLDVISLKEEGLEKIGTWDPASGLNMTESQKGK
PANITDSLSNRSLIVTTILEEPYVLFKKSDKPLYGNDRFEGYCIDLLRELSTILGFTYEI
RLVEDGKYGAQDDVNGQWNGMVRELIDHKADLAVAPLAITYVREKVIDFSKPFMTLGISI
LYRKPNGTNPGVFSFLNPLSPDIWMYILLAYLGVSCVLFVIARFSPYEWYNPHPCNPDSD
VVENNFTLLNSFWFGVGALMQQGSELMPKALSTRIVGGIWWFFTLIIISSYTANLAAFLT
VERMESPIDSADDLAKQTKIEYGAVEDGATMTFFKKSKISTYDKMWAFMSSRRQSVLVKS
NEEGIQRVLTSDYAFLMESTTIEFVTQRNCNLTQIGGLIDSKGYGVGTPMGSPYRDKITI
AILQLQEEGKLHMMKEKWWRGNGCPEEESKEASALGVQNIGGIFIVLAAGLVLSVFVAVG
EFLYKSKKNAQLEKRSFCSAMVEELRMSLKCQRRLKHKPQAPVIVKTEEVINMHTFNDRR
LPGKETMA
|
|
|
|---|
| BDBM50373179 |
|---|
| n/a |
|---|
| Name | BDBM50373179 |
|---|
| Synonyms: | CHEMBL260327 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C5H8N2O4 |
|---|
| Mol. Mass. | 160.128 |
|---|
| SMILES | N[C@H]([C@H]1CC(O)=NO1)C(O)=O |c:5| |
|---|
| Structure | 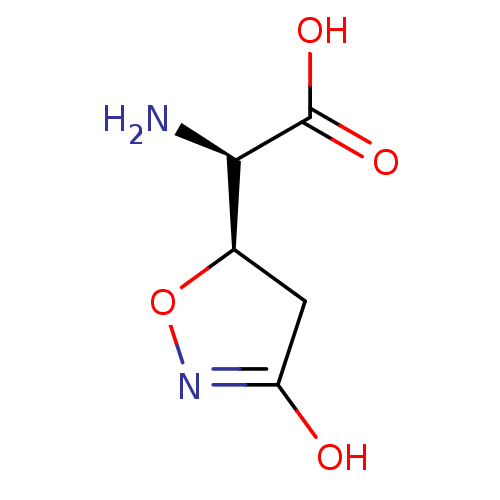
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pinto, A; Conti, P; De Amici, M; Tamborini, L; Madsen, U; Nielsen, B; Christesen, T; Bräuner-Osborne, H; De Micheli, C Synthesis and pharmacological characterization at glutamate receptors of the four enantiopure isomers of tricholomic acid. J Med Chem51:2311-5 (2008) [PubMed] Article
Pinto, A; Conti, P; De Amici, M; Tamborini, L; Madsen, U; Nielsen, B; Christesen, T; Bräuner-Osborne, H; De Micheli, C Synthesis and pharmacological characterization at glutamate receptors of the four enantiopure isomers of tricholomic acid. J Med Chem51:2311-5 (2008) [PubMed] Article