| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Galanin receptor type 2 |
|---|
| Ligand | BDBM50273353 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_561975 (CHEMBL1010038) |
|---|
| Ki | 24±n/a nM |
|---|
| Citation |  Bulaj, G; Green, BR; Lee, HK; Robertson, CR; White, K; Zhang, L; Sochanska, M; Flynn, SP; Scholl, EA; Pruess, TH; Smith, MD; White, HS Design, synthesis, and characterization of high-affinity, systemically-active galanin analogues with potent anticonvulsant activities. J Med Chem51:8038-47 (2008) [PubMed] Article Bulaj, G; Green, BR; Lee, HK; Robertson, CR; White, K; Zhang, L; Sochanska, M; Flynn, SP; Scholl, EA; Pruess, TH; Smith, MD; White, HS Design, synthesis, and characterization of high-affinity, systemically-active galanin analogues with potent anticonvulsant activities. J Med Chem51:8038-47 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Galanin receptor type 2 |
|---|
| Name: | Galanin receptor type 2 |
|---|
| Synonyms: | GALNR2 | GALR2 | GALR2_HUMAN | Galanin R2 | Galanin receptor 2 | Galanin receptor type 2 | Galanin receptor type 2 (GAL2-R) (GALR2). |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 41724.40 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Galanin R2 GALR2 HUMAN::O43603 |
|---|
| Residue: | 387 |
|---|
| Sequence: | MNVSGCPGAGNASQAGGGGGWHPEAVIVPLLFALIFLVGTVGNTLVLAVLLRGGQAVSTT
NLFILNLGVADLCFILCCVPFQATIYTLDGWVFGSLLCKAVHFLIFLTMHASSFTLAAVS
LDRYLAIRYPLHSRELRTPRNALAAIGLIWGLSLLFSGPYLSYYRQSQLANLTVCHPAWS
APRRRAMDICTFVFSYLLPVLVLGLTYARTLRYLWRAVDPVAAGSGARRAKRKVTRMILI
VAALFCLCWMPHHALILCVWFGQFPLTRATYALRILSHLVSYANSCVNPIVYALVSKHFR
KGFRTICAGLLGRAPGRASGRVCAAARGTHSGSVLERESSDLLHMSEAAGALRPCPGASQ
PCILEPCPGPSWQGPKAGDSILTVDVA
|
|
|
|---|
| BDBM50273353 |
|---|
| n/a |
|---|
| Name | BDBM50273353 |
|---|
| Synonyms: | (S)-1-(2-((S)-2-((S)-2-((S)-2-(2-((S)-2-((S)-2-((S)-2-((S)-2-((2S,3R)-2-((S)-3-(1H-indol-3-yl)-2-(2-(methylamino)acetamido)propanamido)-3-hydroxybutanamido)-4-methylpentanamido)-4-amino-4-oxobutanamido)-3-hydroxypropanamido)propanamido)acetamido)-3-(4-hydroxyphenyl)propanamido)-4-methylpentanamido)-4-methylpentanamido)acetyl)-N-((S)-6-amino-1-((S)-6-amino-1-((S)-6-amino-1-((S)-1,6-diamino-1-oxohexan-2-ylamino)-1-oxohexan-2-ylamino)-1-oxohexan-2-ylamino)-1-oxohexan-2-yl)pyrrolidine-2-carboxamide | CHEMBL525055 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C88H144N24O21 |
|---|
| Mol. Mass. | 1874.2332 |
|---|
| SMILES | CNCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(N)=O |r| |
|---|
| Structure | 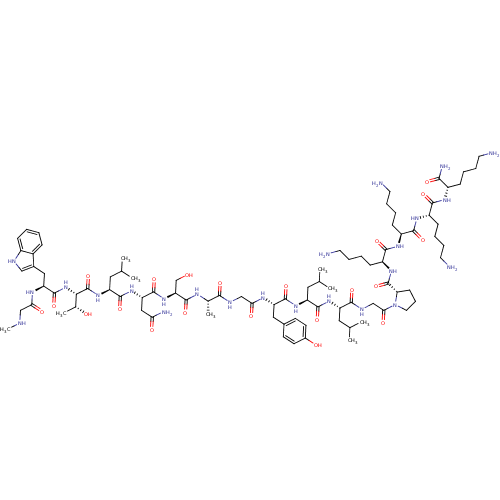
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bulaj, G; Green, BR; Lee, HK; Robertson, CR; White, K; Zhang, L; Sochanska, M; Flynn, SP; Scholl, EA; Pruess, TH; Smith, MD; White, HS Design, synthesis, and characterization of high-affinity, systemically-active galanin analogues with potent anticonvulsant activities. J Med Chem51:8038-47 (2008) [PubMed] Article
Bulaj, G; Green, BR; Lee, HK; Robertson, CR; White, K; Zhang, L; Sochanska, M; Flynn, SP; Scholl, EA; Pruess, TH; Smith, MD; White, HS Design, synthesis, and characterization of high-affinity, systemically-active galanin analogues with potent anticonvulsant activities. J Med Chem51:8038-47 (2008) [PubMed] Article