 Found 27640 hits with Last Name = 'zhang' and Initial = 'l'
Found 27640 hits with Last Name = 'zhang' and Initial = 'l' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207116

(CHEMBL3905247 | US9550741, I-4)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2[nH]1 |r,wU:6.6,wD:3.2,(25.87,-26.54,;26.59,-25.21,;25.82,-23.88,;24.29,-23.88,;23.52,-22.54,;21.97,-22.54,;21.2,-23.88,;19.67,-23.88,;18.9,-22.54,;17.36,-22.54,;16.59,-23.88,;15.06,-23.88,;14.29,-22.54,;15.06,-21.21,;16.59,-21.21,;12.75,-22.54,;11.83,-23.78,;10.4,-23.31,;10.4,-21.77,;9.27,-20.75,;9.58,-19.21,;11.07,-18.75,;12.13,-19.78,;11.83,-21.26,;21.97,-25.21,;23.52,-25.21,;28.13,-25.11,;28.95,-23.83,;30.43,-24.24,;31.72,-23.36,;33.1,-24.08,;33.15,-25.62,;31.87,-26.44,;30.48,-25.77,;29.05,-26.29,)| Show InChI InChI=1S/C28H33N5OS/c34-28(25-19-21-5-1-3-7-24(21)30-25)29-22-11-9-20(10-12-22)13-14-32-15-17-33(18-16-32)27-23-6-2-4-8-26(23)35-31-27/h1-8,19-20,22,30H,9-18H2,(H,29,34)/t20-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| US Patent
| 0.0290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207116

(CHEMBL3905247 | US9550741, I-4)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2[nH]1 |r,wU:6.6,wD:3.2,(25.87,-26.54,;26.59,-25.21,;25.82,-23.88,;24.29,-23.88,;23.52,-22.54,;21.97,-22.54,;21.2,-23.88,;19.67,-23.88,;18.9,-22.54,;17.36,-22.54,;16.59,-23.88,;15.06,-23.88,;14.29,-22.54,;15.06,-21.21,;16.59,-21.21,;12.75,-22.54,;11.83,-23.78,;10.4,-23.31,;10.4,-21.77,;9.27,-20.75,;9.58,-19.21,;11.07,-18.75,;12.13,-19.78,;11.83,-21.26,;21.97,-25.21,;23.52,-25.21,;28.13,-25.11,;28.95,-23.83,;30.43,-24.24,;31.72,-23.36,;33.1,-24.08,;33.15,-25.62,;31.87,-26.44,;30.48,-25.77,;29.05,-26.29,)| Show InChI InChI=1S/C28H33N5OS/c34-28(25-19-21-5-1-3-7-24(21)30-25)29-22-11-9-20(10-12-22)13-14-32-15-17-33(18-16-32)27-23-6-2-4-8-26(23)35-31-27/h1-8,19-20,22,30H,9-18H2,(H,29,34)/t20-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| US Patent
| 0.0290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263372
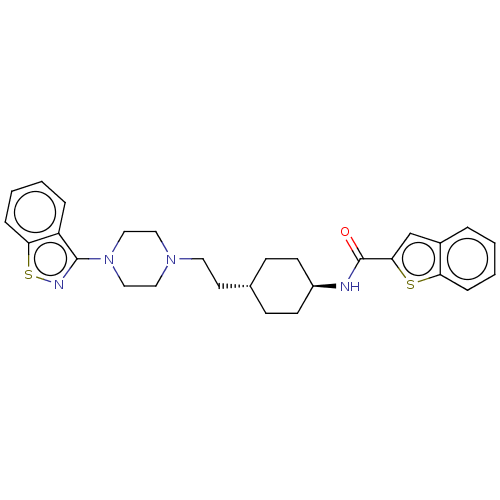
(US9550741, I-6)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2s1 |r,wU:3.2,wD:6.6,(-5.89,-.2,;-5.89,1.34,;-4.56,2.11,;-3.22,1.34,;-1.89,2.11,;-.56,1.34,;-.56,-.2,;.78,-.97,;2.11,-.2,;3.44,-.97,;3.44,-2.51,;4.78,-3.28,;6.11,-2.51,;6.11,-.97,;4.78,-.2,;7.45,-3.28,;7.45,-4.82,;8.91,-5.3,;9.81,-4.05,;11.35,-3.89,;11.97,-2.48,;11.07,-1.24,;9.54,-1.4,;8.91,-2.8,;-1.89,-.97,;-3.22,-.2,;-7.23,2.11,;-7.39,3.64,;-8.89,3.96,;-9.66,5.3,;-11.2,5.3,;-11.97,3.96,;-11.2,2.63,;-9.66,2.63,;-8.63,1.48,)| Show InChI InChI=1S/C28H32N4OS2/c33-28(26-19-21-5-1-3-7-24(21)34-26)29-22-11-9-20(10-12-22)13-14-31-15-17-32(18-16-31)27-23-6-2-4-8-25(23)35-30-27/h1-8,19-20,22H,9-18H2,(H,29,33)/t20-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263372

(US9550741, I-6)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2s1 |r,wU:3.2,wD:6.6,(-5.89,-.2,;-5.89,1.34,;-4.56,2.11,;-3.22,1.34,;-1.89,2.11,;-.56,1.34,;-.56,-.2,;.78,-.97,;2.11,-.2,;3.44,-.97,;3.44,-2.51,;4.78,-3.28,;6.11,-2.51,;6.11,-.97,;4.78,-.2,;7.45,-3.28,;7.45,-4.82,;8.91,-5.3,;9.81,-4.05,;11.35,-3.89,;11.97,-2.48,;11.07,-1.24,;9.54,-1.4,;8.91,-2.8,;-1.89,-.97,;-3.22,-.2,;-7.23,2.11,;-7.39,3.64,;-8.89,3.96,;-9.66,5.3,;-11.2,5.3,;-11.97,3.96,;-11.2,2.63,;-9.66,2.63,;-8.63,1.48,)| Show InChI InChI=1S/C28H32N4OS2/c33-28(26-19-21-5-1-3-7-24(21)34-26)29-22-11-9-20(10-12-22)13-14-31-15-17-32(18-16-31)27-23-6-2-4-8-25(23)35-30-27/h1-8,19-20,22H,9-18H2,(H,29,33)/t20-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207143
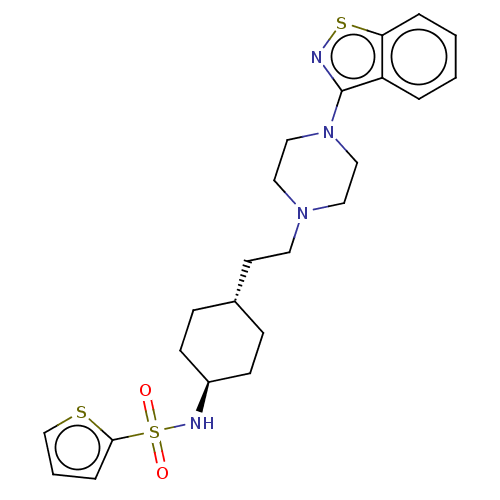
(CHEMBL3966842 | US9550741, II-1)Show SMILES O=S(=O)(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cccs1 |r,wU:4.3,wD:7.7,(11.63,-23.22,;13.07,-23.76,;11.88,-24.73,;13.9,-22.46,;15.43,-22.53,;16.27,-21.24,;17.8,-21.31,;18.51,-22.68,;20.04,-22.75,;20.87,-21.45,;22.41,-21.53,;23.25,-20.23,;24.78,-20.3,;25.49,-21.67,;24.66,-22.97,;23.12,-22.9,;27.03,-21.75,;27.88,-23.03,;29.36,-22.62,;29.43,-21.08,;30.62,-20.11,;30.37,-18.59,;28.93,-18.05,;27.74,-19.02,;27.99,-20.54,;17.68,-23.98,;16.14,-23.9,;13.77,-25.13,;15.29,-25.35,;15.54,-26.87,;14.18,-27.58,;13.08,-26.5,)| Show InChI InChI=1S/C23H30N4O2S3/c28-32(29,22-6-3-17-30-22)25-19-9-7-18(8-10-19)11-12-26-13-15-27(16-14-26)23-20-4-1-2-5-21(20)31-24-23/h1-6,17-19,25H,7-16H2/t18-,19- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207143

(CHEMBL3966842 | US9550741, II-1)Show SMILES O=S(=O)(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cccs1 |r,wU:4.3,wD:7.7,(11.63,-23.22,;13.07,-23.76,;11.88,-24.73,;13.9,-22.46,;15.43,-22.53,;16.27,-21.24,;17.8,-21.31,;18.51,-22.68,;20.04,-22.75,;20.87,-21.45,;22.41,-21.53,;23.25,-20.23,;24.78,-20.3,;25.49,-21.67,;24.66,-22.97,;23.12,-22.9,;27.03,-21.75,;27.88,-23.03,;29.36,-22.62,;29.43,-21.08,;30.62,-20.11,;30.37,-18.59,;28.93,-18.05,;27.74,-19.02,;27.99,-20.54,;17.68,-23.98,;16.14,-23.9,;13.77,-25.13,;15.29,-25.35,;15.54,-26.87,;14.18,-27.58,;13.08,-26.5,)| Show InChI InChI=1S/C23H30N4O2S3/c28-32(29,22-6-3-17-30-22)25-19-9-7-18(8-10-19)11-12-26-13-15-27(16-14-26)23-20-4-1-2-5-21(20)31-24-23/h1-6,17-19,25H,7-16H2/t18-,19- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207094
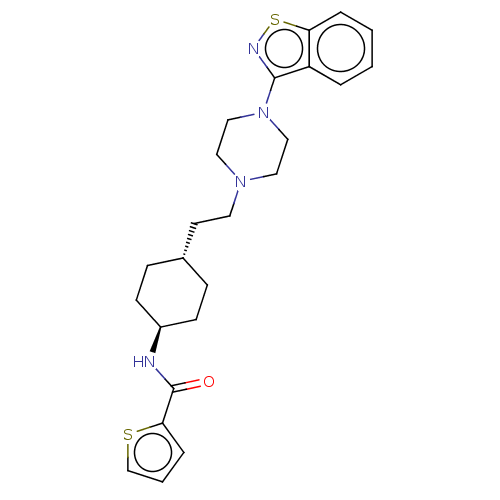
(CHEMBL3976282 | US9550741, I-2)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cccs1 |r,wU:3.2,wD:6.6,(15.49,-22.15,;14.77,-20.77,;15.54,-19.44,;17.08,-19.44,;17.84,-18.11,;19.39,-18.11,;20.15,-19.44,;21.69,-19.44,;22.46,-18.11,;23.99,-18.11,;24.76,-19.44,;26.3,-19.44,;27.06,-18.11,;26.3,-16.77,;24.76,-16.77,;28.61,-18.11,;29.53,-19.33,;31.01,-18.87,;31.01,-17.34,;32.14,-16.31,;31.83,-14.78,;30.34,-14.31,;29.22,-15.34,;29.53,-16.88,;19.39,-20.77,;17.84,-20.77,;13.24,-20.67,;12.42,-19.38,;10.93,-19.79,;10.88,-21.34,;12.32,-21.9,)| Show InChI InChI=1S/C24H30N4OS2/c29-24(22-6-3-17-30-22)25-19-9-7-18(8-10-19)11-12-27-13-15-28(16-14-27)23-20-4-1-2-5-21(20)31-26-23/h1-6,17-19H,7-16H2,(H,25,29)/t18-,19- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207094

(CHEMBL3976282 | US9550741, I-2)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cccs1 |r,wU:3.2,wD:6.6,(15.49,-22.15,;14.77,-20.77,;15.54,-19.44,;17.08,-19.44,;17.84,-18.11,;19.39,-18.11,;20.15,-19.44,;21.69,-19.44,;22.46,-18.11,;23.99,-18.11,;24.76,-19.44,;26.3,-19.44,;27.06,-18.11,;26.3,-16.77,;24.76,-16.77,;28.61,-18.11,;29.53,-19.33,;31.01,-18.87,;31.01,-17.34,;32.14,-16.31,;31.83,-14.78,;30.34,-14.31,;29.22,-15.34,;29.53,-16.88,;19.39,-20.77,;17.84,-20.77,;13.24,-20.67,;12.42,-19.38,;10.93,-19.79,;10.88,-21.34,;12.32,-21.9,)| Show InChI InChI=1S/C24H30N4OS2/c29-24(22-6-3-17-30-22)25-19-9-7-18(8-10-19)11-12-27-13-15-28(16-14-27)23-20-4-1-2-5-21(20)31-26-23/h1-6,17-19H,7-16H2,(H,25,29)/t18-,19- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207158
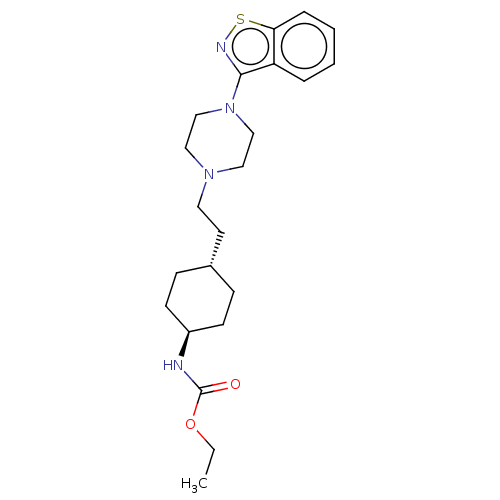
(CHEMBL3982486 | US9550741, III-2)Show SMILES CCOC(=O)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:6.5,wD:9.9,(23.48,-37.39,;25.01,-37.44,;25.84,-36.17,;27.39,-36.27,;28.1,-37.6,;28.15,-34.94,;29.7,-34.94,;30.48,-33.61,;32.01,-33.61,;32.78,-34.95,;34.32,-34.96,;35.09,-33.62,;36.64,-33.62,;37.41,-34.96,;38.95,-34.97,;39.72,-33.63,;38.95,-32.29,;37.42,-32.29,;41.26,-33.64,;42.19,-34.87,;43.56,-34.41,;43.57,-32.88,;44.7,-31.85,;44.39,-30.31,;43.02,-29.84,;41.84,-30.87,;42.19,-32.36,;32.01,-36.28,;30.47,-36.28,)| Show InChI InChI=1S/C22H32N4O2S/c1-2-28-22(27)23-18-9-7-17(8-10-18)11-12-25-13-15-26(16-14-25)21-19-5-3-4-6-20(19)29-24-21/h3-6,17-18H,2,7-16H2,1H3,(H,23,27)/t17-,18- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207158

(CHEMBL3982486 | US9550741, III-2)Show SMILES CCOC(=O)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:6.5,wD:9.9,(23.48,-37.39,;25.01,-37.44,;25.84,-36.17,;27.39,-36.27,;28.1,-37.6,;28.15,-34.94,;29.7,-34.94,;30.48,-33.61,;32.01,-33.61,;32.78,-34.95,;34.32,-34.96,;35.09,-33.62,;36.64,-33.62,;37.41,-34.96,;38.95,-34.97,;39.72,-33.63,;38.95,-32.29,;37.42,-32.29,;41.26,-33.64,;42.19,-34.87,;43.56,-34.41,;43.57,-32.88,;44.7,-31.85,;44.39,-30.31,;43.02,-29.84,;41.84,-30.87,;42.19,-32.36,;32.01,-36.28,;30.47,-36.28,)| Show InChI InChI=1S/C22H32N4O2S/c1-2-28-22(27)23-18-9-7-17(8-10-18)11-12-25-13-15-26(16-14-25)21-19-5-3-4-6-20(19)29-24-21/h3-6,17-18H,2,7-16H2,1H3,(H,23,27)/t17-,18- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263429

(US9550741, III-11)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)OCc1ccccc1 |r,wU:3.2,wD:6.6,(-5.6,.24,;-5.6,1.78,;-4.26,2.55,;-2.93,1.78,;-1.6,2.55,;-.26,1.78,;-.26,.24,;1.07,-.53,;2.4,.24,;3.74,-.53,;3.74,-2.07,;5.07,-2.84,;6.41,-2.07,;6.41,-.53,;5.07,.24,;7.74,-2.84,;7.74,-4.38,;9.2,-4.86,;10.11,-3.61,;11.64,-3.45,;12.27,-2.04,;11.36,-.8,;9.83,-.96,;9.2,-2.37,;-1.6,-.53,;-2.93,.24,;-6.93,2.55,;-8.27,1.78,;-9.6,2.55,;-9.6,4.09,;-10.93,4.86,;-12.27,4.09,;-12.27,2.55,;-10.93,1.78,)| Show InChI InChI=1S/C27H34N4O2S/c32-27(33-20-22-6-2-1-3-7-22)28-23-12-10-21(11-13-23)14-15-30-16-18-31(19-17-30)26-24-8-4-5-9-25(24)34-29-26/h1-9,21,23H,10-20H2,(H,28,32)/t21-,23- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263429

(US9550741, III-11)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)OCc1ccccc1 |r,wU:3.2,wD:6.6,(-5.6,.24,;-5.6,1.78,;-4.26,2.55,;-2.93,1.78,;-1.6,2.55,;-.26,1.78,;-.26,.24,;1.07,-.53,;2.4,.24,;3.74,-.53,;3.74,-2.07,;5.07,-2.84,;6.41,-2.07,;6.41,-.53,;5.07,.24,;7.74,-2.84,;7.74,-4.38,;9.2,-4.86,;10.11,-3.61,;11.64,-3.45,;12.27,-2.04,;11.36,-.8,;9.83,-.96,;9.2,-2.37,;-1.6,-.53,;-2.93,.24,;-6.93,2.55,;-8.27,1.78,;-9.6,2.55,;-9.6,4.09,;-10.93,4.86,;-12.27,4.09,;-12.27,2.55,;-10.93,1.78,)| Show InChI InChI=1S/C27H34N4O2S/c32-27(33-20-22-6-2-1-3-7-22)28-23-12-10-21(11-13-23)14-15-30-16-18-31(19-17-30)26-24-8-4-5-9-25(24)34-29-26/h1-9,21,23H,10-20H2,(H,28,32)/t21-,23- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263393
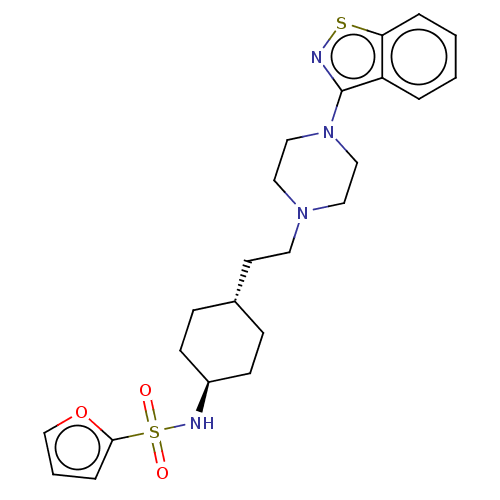
(US9550741, II-3)Show SMILES O=S(=O)(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1ccco1 |r,wU:4.3,wD:7.7,(-7.08,.38,;-7.08,1.92,;-7.08,3.46,;-5.75,2.69,;-4.41,1.92,;-3.08,2.7,;-1.75,1.92,;-1.75,.38,;-.41,-.39,;.92,.38,;2.26,-.39,;2.26,-1.93,;3.59,-2.7,;4.92,-1.93,;4.92,-.39,;3.59,.38,;6.26,-2.7,;6.26,-4.24,;7.72,-4.71,;8.63,-3.47,;10.16,-3.3,;10.78,-1.9,;9.88,-.65,;8.35,-.81,;7.72,-2.22,;-3.08,-.39,;-4.41,.38,;-8.41,2.69,;-8.41,4.23,;-9.88,4.71,;-10.78,3.46,;-9.88,2.22,)| Show InChI InChI=1S/C23H30N4O3S2/c28-32(29,22-6-3-17-30-22)25-19-9-7-18(8-10-19)11-12-26-13-15-27(16-14-26)23-20-4-1-2-5-21(20)31-24-23/h1-6,17-19,25H,7-16H2/t18-,19- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263393

(US9550741, II-3)Show SMILES O=S(=O)(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1ccco1 |r,wU:4.3,wD:7.7,(-7.08,.38,;-7.08,1.92,;-7.08,3.46,;-5.75,2.69,;-4.41,1.92,;-3.08,2.7,;-1.75,1.92,;-1.75,.38,;-.41,-.39,;.92,.38,;2.26,-.39,;2.26,-1.93,;3.59,-2.7,;4.92,-1.93,;4.92,-.39,;3.59,.38,;6.26,-2.7,;6.26,-4.24,;7.72,-4.71,;8.63,-3.47,;10.16,-3.3,;10.78,-1.9,;9.88,-.65,;8.35,-.81,;7.72,-2.22,;-3.08,-.39,;-4.41,.38,;-8.41,2.69,;-8.41,4.23,;-9.88,4.71,;-10.78,3.46,;-9.88,2.22,)| Show InChI InChI=1S/C23H30N4O3S2/c28-32(29,22-6-3-17-30-22)25-19-9-7-18(8-10-19)11-12-26-13-15-27(16-14-26)23-20-4-1-2-5-21(20)31-24-23/h1-6,17-19,25H,7-16H2/t18-,19- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207113
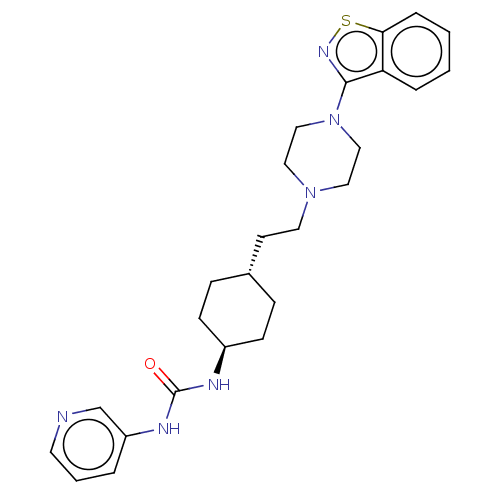
(CHEMBL3896937 | US9550741, IV-3)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)Nc1cccnc1 |r,wU:3.2,wD:6.6,(15.23,-21.5,;14.88,-22.99,;15.9,-24.12,;17.39,-23.82,;17.85,-22.32,;19.4,-22.02,;20.42,-23.15,;21.91,-22.84,;22.94,-23.97,;24.43,-23.66,;24.95,-22.17,;26.43,-21.87,;27.46,-22.99,;27,-24.48,;25.45,-24.79,;28.95,-22.68,;29.62,-21.3,;31.1,-21.45,;31.47,-22.94,;32.8,-23.71,;32.8,-25.25,;31.47,-26.02,;30.13,-25.25,;30.13,-23.71,;19.91,-24.63,;18.42,-24.95,;13.38,-23.35,;12.25,-22.27,;10.76,-22.68,;9.75,-21.6,;10.16,-20.12,;11.58,-19.7,;12.67,-20.78,)| Show InChI InChI=1S/C25H32N6OS/c32-25(28-21-4-3-12-26-18-21)27-20-9-7-19(8-10-20)11-13-30-14-16-31(17-15-30)24-22-5-1-2-6-23(22)33-29-24/h1-6,12,18-20H,7-11,13-17H2,(H2,27,28,32)/t19-,20- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207113

(CHEMBL3896937 | US9550741, IV-3)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)Nc1cccnc1 |r,wU:3.2,wD:6.6,(15.23,-21.5,;14.88,-22.99,;15.9,-24.12,;17.39,-23.82,;17.85,-22.32,;19.4,-22.02,;20.42,-23.15,;21.91,-22.84,;22.94,-23.97,;24.43,-23.66,;24.95,-22.17,;26.43,-21.87,;27.46,-22.99,;27,-24.48,;25.45,-24.79,;28.95,-22.68,;29.62,-21.3,;31.1,-21.45,;31.47,-22.94,;32.8,-23.71,;32.8,-25.25,;31.47,-26.02,;30.13,-25.25,;30.13,-23.71,;19.91,-24.63,;18.42,-24.95,;13.38,-23.35,;12.25,-22.27,;10.76,-22.68,;9.75,-21.6,;10.16,-20.12,;11.58,-19.7,;12.67,-20.78,)| Show InChI InChI=1S/C25H32N6OS/c32-25(28-21-4-3-12-26-18-21)27-20-9-7-19(8-10-20)11-13-30-14-16-31(17-15-30)24-22-5-1-2-6-23(22)33-29-24/h1-6,12,18-20H,7-11,13-17H2,(H2,27,28,32)/t19-,20- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Homo sapiens (Human)) | BDBM21864
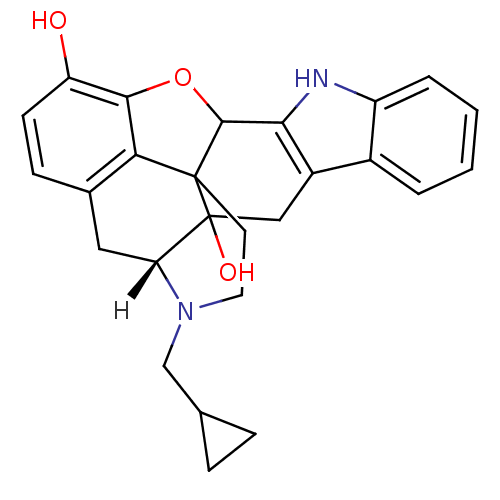
((21R)-22-(cyclopropylmethyl)-14-oxa-11,22-diazahep...)Show SMILES [H][C@@]12Cc3ccc(O)c4OC5c6[nH]c7ccccc7c6CC1(O)C5(CCN2CC1CC1)c34 |THB:27:26:21:31.2.3| Show InChI InChI=1S/C26H26N2O3/c29-19-8-7-15-11-20-26(30)12-17-16-3-1-2-4-18(16)27-22(17)24-25(26,21(15)23(19)31-24)9-10-28(20)13-14-5-6-14/h1-4,7-8,14,20,24,27,29-30H,5-6,9-13H2/t20-,24?,25?,26?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
[3H]-Cl-DPDPE competition binding assay using human cloned opioid receptor delta 1 |
J Med Chem 47: 281-4 (2004)
Article DOI: 10.1021/jm030419a
BindingDB Entry DOI: 10.7270/Q20K2990 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263388

(US9550741, I-22)Show SMILES O=C(N[C@@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2[nH]1 |r,wU:3.2,6.6,(-5.89,-.2,;-5.89,1.34,;-4.56,2.11,;-3.22,1.34,;-3.22,-.2,;-1.89,-.97,;-.56,-.2,;.78,-.97,;2.11,-.2,;3.44,-.97,;3.44,-2.51,;4.78,-3.28,;6.11,-2.51,;6.11,-.97,;4.78,-.2,;7.45,-3.28,;7.45,-4.82,;8.91,-5.3,;9.81,-4.05,;11.35,-3.89,;11.97,-2.48,;11.07,-1.24,;9.54,-1.4,;8.91,-2.8,;-.56,1.34,;-1.89,2.11,;-7.23,2.11,;-7.39,3.64,;-8.89,3.96,;-9.66,5.3,;-11.2,5.3,;-11.97,3.96,;-11.2,2.63,;-9.66,2.63,;-8.63,1.48,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0710 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263388

(US9550741, I-22)Show SMILES O=C(N[C@@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2[nH]1 |r,wU:3.2,6.6,(-5.89,-.2,;-5.89,1.34,;-4.56,2.11,;-3.22,1.34,;-3.22,-.2,;-1.89,-.97,;-.56,-.2,;.78,-.97,;2.11,-.2,;3.44,-.97,;3.44,-2.51,;4.78,-3.28,;6.11,-2.51,;6.11,-.97,;4.78,-.2,;7.45,-3.28,;7.45,-4.82,;8.91,-5.3,;9.81,-4.05,;11.35,-3.89,;11.97,-2.48,;11.07,-1.24,;9.54,-1.4,;8.91,-2.8,;-.56,1.34,;-1.89,2.11,;-7.23,2.11,;-7.39,3.64,;-8.89,3.96,;-9.66,5.3,;-11.2,5.3,;-11.97,3.96,;-11.2,2.63,;-9.66,2.63,;-8.63,1.48,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0710 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50090002
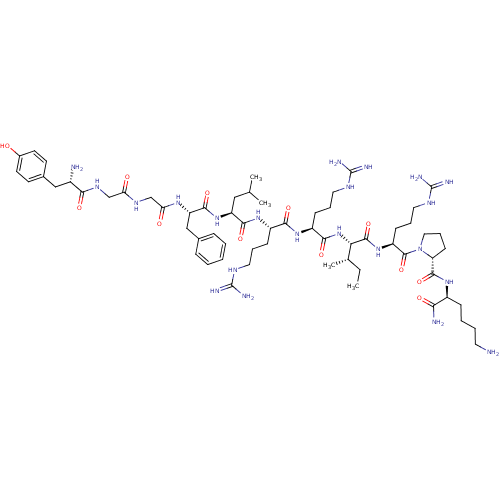
(CHEMBL411003 | Dynorphin A (1-11)-NH2H-Tyr-Gly-Gly...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCCN)C(N)=O Show InChI InChI=1S/C63H104N22O12/c1-5-37(4)51(59(96)82-45(20-13-29-75-63(71)72)60(97)85-30-14-21-48(85)58(95)79-42(52(66)89)17-9-10-26-64)84-55(92)44(19-12-28-74-62(69)70)80-54(91)43(18-11-27-73-61(67)68)81-56(93)46(31-36(2)3)83-57(94)47(33-38-15-7-6-8-16-38)78-50(88)35-76-49(87)34-77-53(90)41(65)32-39-22-24-40(86)25-23-39/h6-8,15-16,22-25,36-37,41-48,51,86H,5,9-14,17-21,26-35,64-65H2,1-4H3,(H2,66,89)(H,76,87)(H,77,90)(H,78,88)(H,79,95)(H,80,91)(H,81,93)(H,82,96)(H,83,94)(H,84,92)(H4,67,68,73)(H4,69,70,74)(H4,71,72,75)/t37-,41-,42-,43-,44-,45-,46-,47-,48+,51-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
Binding affinity towards cloned human Opioid receptor kappa 1 in CHO cell membranes. |
J Med Chem 43: 2698-702 (2000)
BindingDB Entry DOI: 10.7270/Q25D8SJ1 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263371
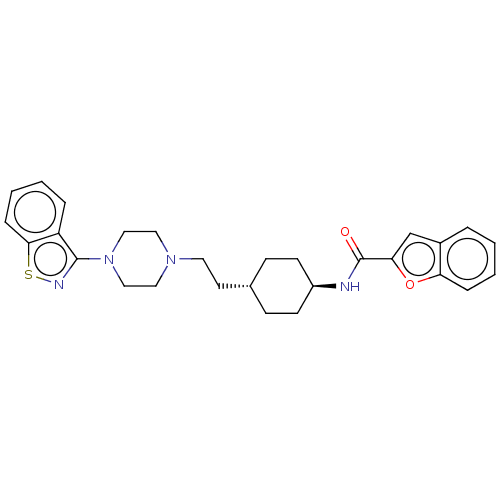
(US9550741, I-5)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2o1 |r,wU:3.2,wD:6.6,(-5.89,-.2,;-5.89,1.34,;-4.56,2.11,;-3.22,1.34,;-1.89,2.11,;-.56,1.34,;-.56,-.2,;.78,-.97,;2.11,-.2,;3.44,-.97,;3.44,-2.51,;4.78,-3.28,;6.11,-2.51,;6.11,-.97,;4.78,-.2,;7.45,-3.28,;7.45,-4.82,;8.91,-5.3,;9.81,-4.05,;11.35,-3.89,;11.97,-2.48,;11.07,-1.24,;9.54,-1.4,;8.91,-2.8,;-1.89,-.97,;-3.22,-.2,;-7.23,2.11,;-7.39,3.64,;-8.89,3.96,;-9.66,5.3,;-11.2,5.3,;-11.97,3.96,;-11.2,2.63,;-9.66,2.63,;-8.63,1.48,)| Show InChI InChI=1S/C28H32N4O2S/c33-28(25-19-21-5-1-3-7-24(21)34-25)29-22-11-9-20(10-12-22)13-14-31-15-17-32(18-16-31)27-23-6-2-4-8-26(23)35-30-27/h1-8,19-20,22H,9-18H2,(H,29,33)/t20-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263371

(US9550741, I-5)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2o1 |r,wU:3.2,wD:6.6,(-5.89,-.2,;-5.89,1.34,;-4.56,2.11,;-3.22,1.34,;-1.89,2.11,;-.56,1.34,;-.56,-.2,;.78,-.97,;2.11,-.2,;3.44,-.97,;3.44,-2.51,;4.78,-3.28,;6.11,-2.51,;6.11,-.97,;4.78,-.2,;7.45,-3.28,;7.45,-4.82,;8.91,-5.3,;9.81,-4.05,;11.35,-3.89,;11.97,-2.48,;11.07,-1.24,;9.54,-1.4,;8.91,-2.8,;-1.89,-.97,;-3.22,-.2,;-7.23,2.11,;-7.39,3.64,;-8.89,3.96,;-9.66,5.3,;-11.2,5.3,;-11.97,3.96,;-11.2,2.63,;-9.66,2.63,;-8.63,1.48,)| Show InChI InChI=1S/C28H32N4O2S/c33-28(25-19-21-5-1-3-7-24(21)34-25)29-22-11-9-20(10-12-22)13-14-31-15-17-32(18-16-31)27-23-6-2-4-8-26(23)35-30-27/h1-8,19-20,22H,9-18H2,(H,29,33)/t20-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263395
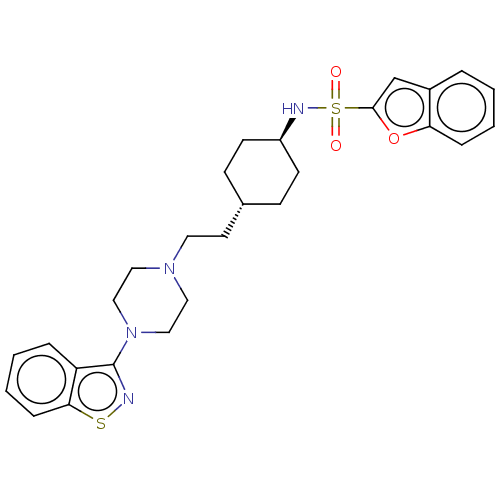
(US9550741, II-5)Show SMILES O=S(=O)(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2o1 |r,wU:4.3,wD:7.7,(-5.89,-.2,;-5.89,1.34,;-5.89,2.88,;-4.56,2.11,;-3.22,1.34,;-1.89,2.11,;-.56,1.34,;-.56,-.2,;.78,-.97,;2.11,-.2,;3.44,-.97,;3.44,-2.51,;4.78,-3.28,;6.11,-2.51,;6.11,-.97,;4.78,-.2,;7.45,-3.28,;7.45,-4.82,;8.91,-5.3,;9.81,-4.05,;11.35,-3.89,;11.97,-2.48,;11.07,-1.24,;9.54,-1.4,;8.91,-2.8,;-1.89,-.97,;-3.22,-.2,;-7.23,2.11,;-7.39,3.64,;-8.89,3.96,;-9.66,5.3,;-11.2,5.3,;-11.97,3.96,;-11.2,2.63,;-9.66,2.63,;-8.63,1.48,)| Show InChI InChI=1S/C27H32N4O3S2/c32-36(33,26-19-21-5-1-3-7-24(21)34-26)29-22-11-9-20(10-12-22)13-14-30-15-17-31(18-16-30)27-23-6-2-4-8-25(23)35-28-27/h1-8,19-20,22,29H,9-18H2/t20-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263395

(US9550741, II-5)Show SMILES O=S(=O)(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cc2ccccc2o1 |r,wU:4.3,wD:7.7,(-5.89,-.2,;-5.89,1.34,;-5.89,2.88,;-4.56,2.11,;-3.22,1.34,;-1.89,2.11,;-.56,1.34,;-.56,-.2,;.78,-.97,;2.11,-.2,;3.44,-.97,;3.44,-2.51,;4.78,-3.28,;6.11,-2.51,;6.11,-.97,;4.78,-.2,;7.45,-3.28,;7.45,-4.82,;8.91,-5.3,;9.81,-4.05,;11.35,-3.89,;11.97,-2.48,;11.07,-1.24,;9.54,-1.4,;8.91,-2.8,;-1.89,-.97,;-3.22,-.2,;-7.23,2.11,;-7.39,3.64,;-8.89,3.96,;-9.66,5.3,;-11.2,5.3,;-11.97,3.96,;-11.2,2.63,;-9.66,2.63,;-8.63,1.48,)| Show InChI InChI=1S/C27H32N4O3S2/c32-36(33,26-19-21-5-1-3-7-24(21)34-26)29-22-11-9-20(10-12-22)13-14-30-15-17-31(18-16-30)27-23-6-2-4-8-25(23)35-28-27/h1-8,19-20,22,29H,9-18H2/t20-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263432

(US9550741, III-14)Show SMILES COC(=O)N[C@@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:5.4,8.8,(-10.27,2.93,;-8.93,3.7,;-7.6,2.93,;-7.6,1.39,;-6.26,3.7,;-4.93,2.93,;-4.93,1.39,;-3.6,.62,;-2.26,1.39,;-.93,.62,;.4,1.39,;1.74,.62,;1.74,-.92,;3.07,-1.69,;4.4,-.92,;4.4,.62,;3.07,1.39,;5.74,-1.69,;5.74,-3.23,;7.2,-3.7,;8.11,-2.46,;9.64,-2.3,;10.27,-.89,;9.36,.36,;7.83,.2,;7.2,-1.21,;-2.26,2.93,;-3.6,3.7,)| Show InChI InChI=1S/C21H30N4O2S/c1-27-21(26)22-17-8-6-16(7-9-17)10-11-24-12-14-25(15-13-24)20-18-4-2-3-5-19(18)28-23-20/h2-5,16-17H,6-15H2,1H3,(H,22,26)/t16-,17+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263432

(US9550741, III-14)Show SMILES COC(=O)N[C@@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:5.4,8.8,(-10.27,2.93,;-8.93,3.7,;-7.6,2.93,;-7.6,1.39,;-6.26,3.7,;-4.93,2.93,;-4.93,1.39,;-3.6,.62,;-2.26,1.39,;-.93,.62,;.4,1.39,;1.74,.62,;1.74,-.92,;3.07,-1.69,;4.4,-.92,;4.4,.62,;3.07,1.39,;5.74,-1.69,;5.74,-3.23,;7.2,-3.7,;8.11,-2.46,;9.64,-2.3,;10.27,-.89,;9.36,.36,;7.83,.2,;7.2,-1.21,;-2.26,2.93,;-3.6,3.7,)| Show InChI InChI=1S/C21H30N4O2S/c1-27-21(26)22-17-8-6-16(7-9-17)10-11-24-12-14-25(15-13-24)20-18-4-2-3-5-19(18)28-23-20/h2-5,16-17H,6-15H2,1H3,(H,22,26)/t16-,17+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Rattus norvegicus) | BDBM50165931
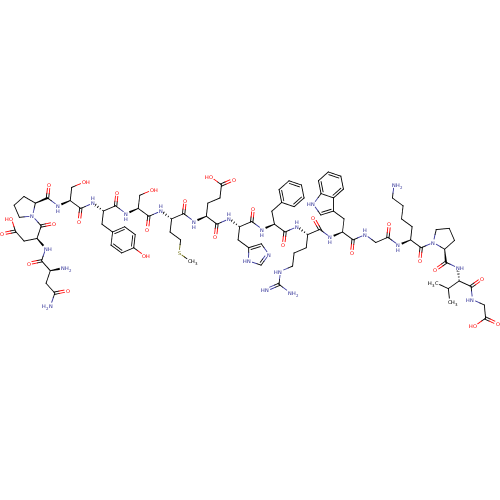
(CHEMBL415165 | NDP-SYSMEHFRWGKPVG)Show SMILES CSCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(=O)NCC(O)=O Show InChI InChI=1S/C90H127N25O26S/c1-47(2)74(87(139)100-43-73(125)126)113-86(138)68-21-13-31-114(68)88(140)59(18-9-10-29-91)102-70(120)42-99-76(128)62(36-50-40-98-55-17-8-7-16-53(50)55)108-77(129)56(19-11-30-97-90(94)95)103-80(132)60(34-48-14-5-4-6-15-48)106-82(134)63(37-51-41-96-46-101-51)109-78(130)57(26-27-71(121)122)104-79(131)58(28-33-142-3)105-83(135)65(44-116)111-81(133)61(35-49-22-24-52(118)25-23-49)107-84(136)66(45-117)112-85(137)67-20-12-32-115(67)89(141)64(39-72(123)124)110-75(127)54(92)38-69(93)119/h4-8,14-17,22-25,40-41,46-47,54,56-68,74,98,116-118H,9-13,18-21,26-39,42-45,91-92H2,1-3H3,(H2,93,119)(H,96,101)(H,99,128)(H,100,139)(H,102,120)(H,103,132)(H,104,131)(H,105,135)(H,106,134)(H,107,136)(H,108,129)(H,109,130)(H,110,127)(H,111,133)(H,112,137)(H,113,138)(H,121,122)(H,123,124)(H,125,126)(H4,94,95,97)/t54-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,74-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity for rat melanocortin-4 receptor |
J Med Chem 48: 3095-8 (2005)
Article DOI: 10.1021/jm0501432
BindingDB Entry DOI: 10.7270/Q2251HQZ |
More data for this
Ligand-Target Pair | |
Melanocyte-stimulating hormone receptor
(Homo sapiens (Human)) | BDBM50165935
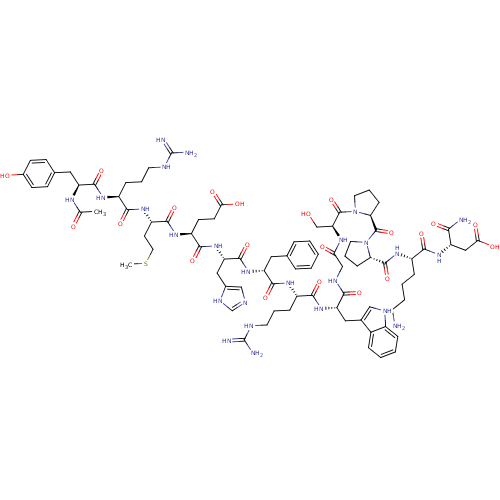
(Ac-YRMEHdFRWGSPPKD-NH2 | CHEMBL414718)Show SMILES CSCC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc1ccc(O)cc1)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CO)C(=O)N1CCC[C@H]1C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(N)=O Show InChI InChI=1S/C84H119N25O21S/c1-46(111)97-60(37-48-23-25-51(112)26-24-48)77(125)99-55(19-10-31-92-83(87)88)72(120)102-58(29-35-131-2)76(124)101-57(27-28-68(114)115)75(123)107-63(39-50-42-91-45-96-50)79(127)105-61(36-47-14-4-3-5-15-47)78(126)100-56(20-11-32-93-84(89)90)74(122)106-62(38-49-41-94-53-17-7-6-16-52(49)53)71(119)95-43-67(113)98-64(44-110)81(129)109-34-13-22-66(109)82(130)108-33-12-21-65(108)80(128)103-54(18-8-9-30-85)73(121)104-59(70(86)118)40-69(116)117/h3-7,14-17,23-26,41-42,45,54-66,94,110,112H,8-13,18-22,27-40,43-44,85H2,1-2H3,(H2,86,118)(H,91,96)(H,95,119)(H,97,111)(H,98,113)(H,99,125)(H,100,126)(H,101,124)(H,102,120)(H,103,128)(H,104,121)(H,105,127)(H,106,122)(H,107,123)(H,114,115)(H,116,117)(H4,87,88,92)(H4,89,90,93)/t54-,55-,56-,57-,58-,59-,60-,61+,62-,63-,64-,65-,66-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of [125I]NDP-alpha-MSH binding to melanocortin-1 receptor expressed in HEK293 cells |
J Med Chem 48: 3095-8 (2005)
Article DOI: 10.1021/jm0501432
BindingDB Entry DOI: 10.7270/Q2251HQZ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50343274
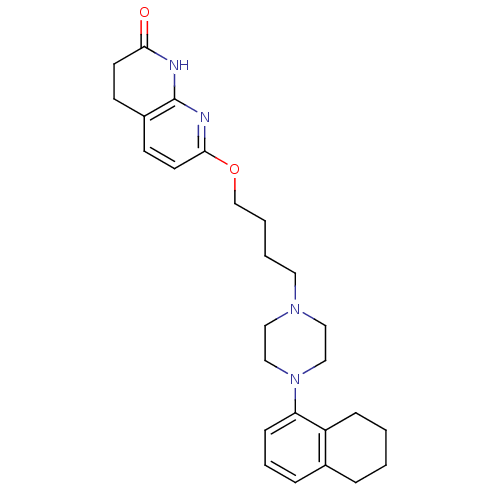
(7-(4-(4-(5,6,7,8-tetrahydronaphthalen-1-yl)piperaz...)Show SMILES O=C1CCc2ccc(OCCCCN3CCN(CC3)c3cccc4CCCCc34)nc2N1 Show InChI InChI=1S/C26H34N4O2/c31-24-12-10-21-11-13-25(28-26(21)27-24)32-19-4-3-14-29-15-17-30(18-16-29)23-9-5-7-20-6-1-2-8-22(20)23/h5,7,9,11,13H,1-4,6,8,10,12,14-19H2,(H,27,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to human 5HT2A receptor |
Bioorg Med Chem Lett 21: 2621-5 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.059
BindingDB Entry DOI: 10.7270/Q29K4BJ3 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263449

(US9550741, RGH-188)Show SMILES CN(C)C(=O)NC1CCC(CCN2CCN(CC2)c2cccc(Cl)c2Cl)CC1 |(,-4.62,;1.33,-5.39,;2.67,-4.62,;1.33,-6.93,;,-7.7,;2.67,-7.7,;2.67,-9.24,;4,-10.01,;4,-11.55,;2.67,-12.32,;2.67,-13.86,;4,-14.63,;4,-16.17,;5.33,-16.94,;5.33,-18.48,;4,-19.25,;2.67,-18.48,;2.67,-16.94,;4,-20.79,;2.67,-21.56,;2.67,-23.1,;4,-23.87,;5.33,-23.1,;6.67,-23.87,;5.33,-21.56,;6.67,-20.79,;1.33,-11.55,;1.33,-10.01,)| Show InChI InChI=1S/C21H32Cl2N4O/c1-25(2)21(28)24-17-8-6-16(7-9-17)10-11-26-12-14-27(15-13-26)19-5-3-4-18(22)20(19)23/h3-5,16-17H,6-15H2,1-2H3,(H,24,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| US Patent
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263421
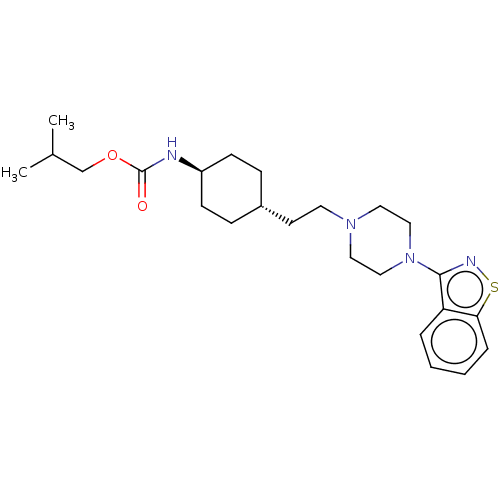
(US9550741, III-3)Show SMILES CC(C)COC(=O)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:8.7,wD:11.11,(-11.6,2.16,;-10.27,2.93,;-10.27,4.47,;-8.93,2.16,;-7.6,2.93,;-6.26,2.16,;-6.26,.62,;-4.93,2.93,;-3.6,2.16,;-2.26,2.93,;-.93,2.16,;-.93,.62,;.4,-.15,;1.74,.62,;3.07,-.15,;3.07,-1.69,;4.4,-2.46,;5.74,-1.69,;5.74,-.15,;4.4,.62,;7.07,-2.46,;7.07,-4,;8.54,-4.47,;9.44,-3.23,;10.97,-3.07,;11.6,-1.66,;10.69,-.41,;9.16,-.57,;8.54,-1.98,;-2.26,-.15,;-3.6,.62,)| Show InChI InChI=1S/C24H36N4O2S/c1-18(2)17-30-24(29)25-20-9-7-19(8-10-20)11-12-27-13-15-28(16-14-27)23-21-5-3-4-6-22(21)31-26-23/h3-6,18-20H,7-17H2,1-2H3,(H,25,29)/t19-,20- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263412
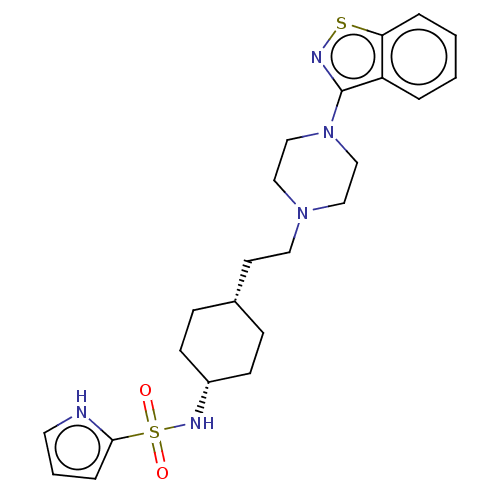
(US9550741, II-17)Show SMILES O=S(=O)(N[C@@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1ccc[nH]1 |r,wU:4.3,7.7,(-7.08,.38,;-7.08,1.92,;-7.08,3.46,;-5.75,2.69,;-4.41,1.92,;-4.41,.38,;-3.08,-.39,;-1.75,.38,;-.41,-.39,;.92,.38,;2.26,-.39,;2.26,-1.93,;3.59,-2.7,;4.92,-1.93,;4.92,-.39,;3.59,.38,;6.26,-2.7,;6.26,-4.24,;7.72,-4.71,;8.63,-3.47,;10.16,-3.3,;10.78,-1.9,;9.88,-.65,;8.35,-.81,;7.72,-2.22,;-1.75,1.92,;-3.08,2.7,;-8.41,2.69,;-8.41,4.23,;-9.88,4.71,;-10.78,3.46,;-9.88,2.22,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263412

(US9550741, II-17)Show SMILES O=S(=O)(N[C@@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1ccc[nH]1 |r,wU:4.3,7.7,(-7.08,.38,;-7.08,1.92,;-7.08,3.46,;-5.75,2.69,;-4.41,1.92,;-4.41,.38,;-3.08,-.39,;-1.75,.38,;-.41,-.39,;.92,.38,;2.26,-.39,;2.26,-1.93,;3.59,-2.7,;4.92,-1.93,;4.92,-.39,;3.59,.38,;6.26,-2.7,;6.26,-4.24,;7.72,-4.71,;8.63,-3.47,;10.16,-3.3,;10.78,-1.9,;9.88,-.65,;8.35,-.81,;7.72,-2.22,;-1.75,1.92,;-3.08,2.7,;-8.41,2.69,;-8.41,4.23,;-9.88,4.71,;-10.78,3.46,;-9.88,2.22,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263421

(US9550741, III-3)Show SMILES CC(C)COC(=O)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:8.7,wD:11.11,(-11.6,2.16,;-10.27,2.93,;-10.27,4.47,;-8.93,2.16,;-7.6,2.93,;-6.26,2.16,;-6.26,.62,;-4.93,2.93,;-3.6,2.16,;-2.26,2.93,;-.93,2.16,;-.93,.62,;.4,-.15,;1.74,.62,;3.07,-.15,;3.07,-1.69,;4.4,-2.46,;5.74,-1.69,;5.74,-.15,;4.4,.62,;7.07,-2.46,;7.07,-4,;8.54,-4.47,;9.44,-3.23,;10.97,-3.07,;11.6,-1.66,;10.69,-.41,;9.16,-.57,;8.54,-1.98,;-2.26,-.15,;-3.6,.62,)| Show InChI InChI=1S/C24H36N4O2S/c1-18(2)17-30-24(29)25-20-9-7-19(8-10-20)11-12-27-13-15-28(16-14-27)23-21-5-3-4-6-22(21)31-26-23/h3-6,18-20H,7-17H2,1-2H3,(H,25,29)/t19-,20- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.0930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Melanocyte-stimulating hormone receptor
(Homo sapiens (Human)) | BDBM50165931

(CHEMBL415165 | NDP-SYSMEHFRWGKPVG)Show SMILES CSCC[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CO)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(=O)NCC(O)=O Show InChI InChI=1S/C90H127N25O26S/c1-47(2)74(87(139)100-43-73(125)126)113-86(138)68-21-13-31-114(68)88(140)59(18-9-10-29-91)102-70(120)42-99-76(128)62(36-50-40-98-55-17-8-7-16-53(50)55)108-77(129)56(19-11-30-97-90(94)95)103-80(132)60(34-48-14-5-4-6-15-48)106-82(134)63(37-51-41-96-46-101-51)109-78(130)57(26-27-71(121)122)104-79(131)58(28-33-142-3)105-83(135)65(44-116)111-81(133)61(35-49-22-24-52(118)25-23-49)107-84(136)66(45-117)112-85(137)67-20-12-32-115(67)89(141)64(39-72(123)124)110-75(127)54(92)38-69(93)119/h4-8,14-17,22-25,40-41,46-47,54,56-68,74,98,116-118H,9-13,18-21,26-39,42-45,91-92H2,1-3H3,(H2,93,119)(H,96,101)(H,99,128)(H,100,139)(H,102,120)(H,103,132)(H,104,131)(H,105,135)(H,106,134)(H,107,136)(H,108,129)(H,109,130)(H,110,127)(H,111,133)(H,112,137)(H,113,138)(H,121,122)(H,123,124)(H,125,126)(H4,94,95,97)/t54-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,74-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of [125I]NDP-alpha-MSH binding to melanocortin-1 receptor expressed in HEK293 cells |
J Med Chem 48: 3095-8 (2005)
Article DOI: 10.1021/jm0501432
BindingDB Entry DOI: 10.7270/Q2251HQZ |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263383
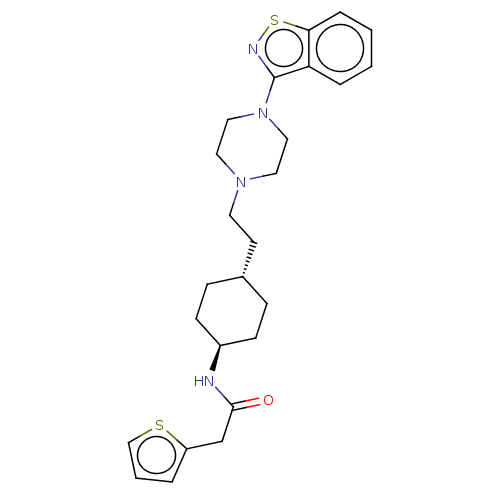
(US9550741, I-17)Show SMILES O=C(Cc1cccs1)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:9.9,wD:12.13,(-6.35,1.05,;-6.35,2.59,;-7.69,3.36,;-9.02,2.59,;-9.5,4.05,;-11.04,4.05,;-11.51,2.59,;-10.27,1.68,;-5.02,3.36,;-3.69,2.59,;-2.35,3.36,;-1.02,2.59,;-1.02,1.05,;.32,.28,;1.65,1.05,;2.98,.28,;2.98,-1.26,;4.32,-2.03,;5.65,-1.26,;5.65,.28,;4.32,1.05,;6.98,-2.03,;6.98,-3.57,;8.45,-4.05,;9.35,-2.8,;10.89,-2.64,;11.51,-1.24,;10.61,.01,;9.08,-.15,;8.45,-1.56,;-2.35,.28,;-3.69,1.05,)| Show InChI InChI=1S/C25H32N4OS2/c30-24(18-21-4-3-17-31-21)26-20-9-7-19(8-10-20)11-12-28-13-15-29(16-14-28)25-22-5-1-2-6-23(22)32-27-25/h1-6,17,19-20H,7-16,18H2,(H,26,30)/t19-,20- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263383

(US9550741, I-17)Show SMILES O=C(Cc1cccs1)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:9.9,wD:12.13,(-6.35,1.05,;-6.35,2.59,;-7.69,3.36,;-9.02,2.59,;-9.5,4.05,;-11.04,4.05,;-11.51,2.59,;-10.27,1.68,;-5.02,3.36,;-3.69,2.59,;-2.35,3.36,;-1.02,2.59,;-1.02,1.05,;.32,.28,;1.65,1.05,;2.98,.28,;2.98,-1.26,;4.32,-2.03,;5.65,-1.26,;5.65,.28,;4.32,1.05,;6.98,-2.03,;6.98,-3.57,;8.45,-4.05,;9.35,-2.8,;10.89,-2.64,;11.51,-1.24,;10.61,.01,;9.08,-.15,;8.45,-1.56,;-2.35,.28,;-3.69,1.05,)| Show InChI InChI=1S/C25H32N4OS2/c30-24(18-21-4-3-17-31-21)26-20-9-7-19(8-10-20)11-12-28-13-15-29(16-14-28)25-22-5-1-2-6-23(22)32-27-25/h1-6,17,19-20H,7-16,18H2,(H,26,30)/t19-,20- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263419
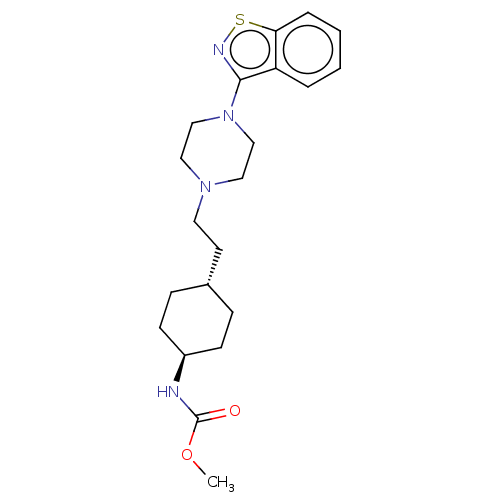
(US9550741, III-1)Show SMILES COC(=O)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:5.4,wD:8.8,(-10.27,2.93,;-8.93,3.7,;-7.6,2.93,;-7.6,1.39,;-6.26,3.7,;-4.93,2.93,;-3.6,3.7,;-2.26,2.93,;-2.26,1.39,;-.93,.62,;.4,1.39,;1.74,.62,;1.74,-.92,;3.07,-1.69,;4.4,-.92,;4.4,.62,;3.07,1.39,;5.74,-1.69,;5.74,-3.23,;7.2,-3.7,;8.11,-2.46,;9.64,-2.3,;10.27,-.89,;9.36,.36,;7.83,.2,;7.2,-1.21,;-3.6,.62,;-4.93,1.39,)| Show InChI InChI=1S/C21H30N4O2S/c1-27-21(26)22-17-8-6-16(7-9-17)10-11-24-12-14-25(15-13-24)20-18-4-2-3-5-19(18)28-23-20/h2-5,16-17H,6-15H2,1H3,(H,22,26)/t16-,17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM263419

(US9550741, III-1)Show SMILES COC(=O)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1 |r,wU:5.4,wD:8.8,(-10.27,2.93,;-8.93,3.7,;-7.6,2.93,;-7.6,1.39,;-6.26,3.7,;-4.93,2.93,;-3.6,3.7,;-2.26,2.93,;-2.26,1.39,;-.93,.62,;.4,1.39,;1.74,.62,;1.74,-.92,;3.07,-1.69,;4.4,-.92,;4.4,.62,;3.07,1.39,;5.74,-1.69,;5.74,-3.23,;7.2,-3.7,;8.11,-2.46,;9.64,-2.3,;10.27,-.89,;9.36,.36,;7.83,.2,;7.2,-1.21,;-3.6,.62,;-4.93,1.39,)| Show InChI InChI=1S/C21H30N4O2S/c1-27-21(26)22-17-8-6-16(7-9-17)10-11-24-12-14-25(15-13-24)20-18-4-2-3-5-19(18)28-23-20/h2-5,16-17H,6-15H2,1H3,(H,22,26)/t16-,17- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 3
(Homo sapiens (Human)) | BDBM50165935

(Ac-YRMEHdFRWGSPPKD-NH2 | CHEMBL414718)Show SMILES CSCC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc1ccc(O)cc1)NC(C)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCC(=O)N[C@@H](CO)C(=O)N1CCC[C@H]1C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(N)=O Show InChI InChI=1S/C84H119N25O21S/c1-46(111)97-60(37-48-23-25-51(112)26-24-48)77(125)99-55(19-10-31-92-83(87)88)72(120)102-58(29-35-131-2)76(124)101-57(27-28-68(114)115)75(123)107-63(39-50-42-91-45-96-50)79(127)105-61(36-47-14-4-3-5-15-47)78(126)100-56(20-11-32-93-84(89)90)74(122)106-62(38-49-41-94-53-17-7-6-16-52(49)53)71(119)95-43-67(113)98-64(44-110)81(129)109-34-13-22-66(109)82(130)108-33-12-21-65(108)80(128)103-54(18-8-9-30-85)73(121)104-59(70(86)118)40-69(116)117/h3-7,14-17,23-26,41-42,45,54-66,94,110,112H,8-13,18-22,27-40,43-44,85H2,1-2H3,(H2,86,118)(H,91,96)(H,95,119)(H,97,111)(H,98,113)(H,99,125)(H,100,126)(H,101,124)(H,102,120)(H,103,128)(H,104,121)(H,105,127)(H,106,122)(H,107,123)(H,114,115)(H,116,117)(H4,87,88,92)(H4,89,90,93)/t54-,55-,56-,57-,58-,59-,60-,61+,62-,63-,64-,65-,66-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of [125I]-NDP-alpha-MSH binding to melanocortin-3 receptor expressed in HEK293 cells |
J Med Chem 48: 3095-8 (2005)
Article DOI: 10.1021/jm0501432
BindingDB Entry DOI: 10.7270/Q2251HQZ |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50029085
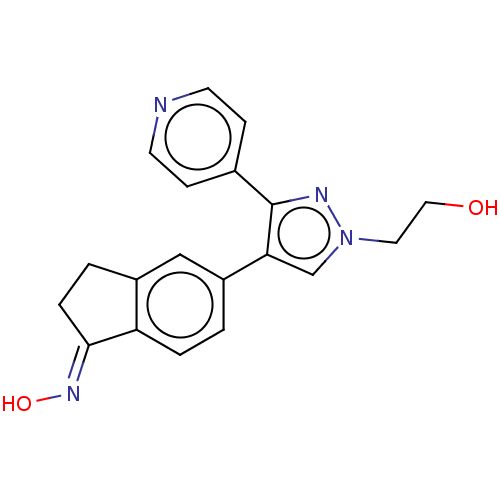
(CHEBI:83405 | CHEMBL525191 | GDC-0879)Show SMILES OCCn1cc(c(n1)-c1ccncc1)-c1ccc2\C(CCc2c1)=N\O Show InChI InChI=1S/C19H18N4O2/c24-10-9-23-12-17(19(21-23)13-5-7-20-8-6-13)15-1-3-16-14(11-15)2-4-18(16)22-25/h1,3,5-8,11-12,24-25H,2,4,9-10H2/b22-18+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nanjing University
Curated by ChEMBL
| Assay Description
Inhibition of BRAF V600E mutant (unknown origin) |
Bioorg Med Chem 22: 6201-8 (2014)
Article DOI: 10.1016/j.bmc.2014.08.029
BindingDB Entry DOI: 10.7270/Q2RB7658 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207091
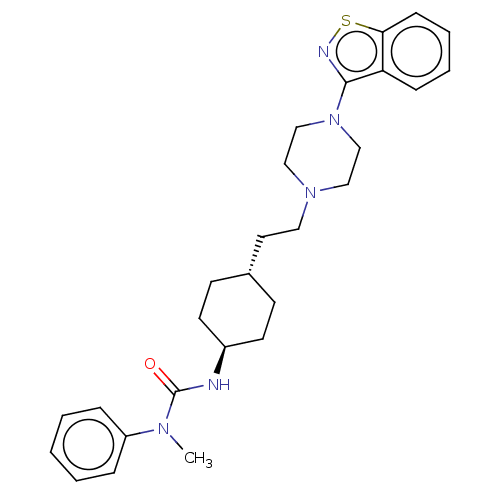
(CHEMBL3953773 | US9550741, IV-6)Show SMILES CN(C(=O)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1ccccc1 |r,wU:5.4,wD:8.8,(19.37,-25.45,;20.19,-24.17,;21.74,-24.22,;22.46,-25.6,;22.51,-22.88,;24.05,-22.88,;24.83,-21.54,;26.37,-21.54,;27.14,-22.88,;28.69,-22.88,;29.46,-21.54,;31,-21.54,;31.77,-22.88,;33.32,-22.88,;34.09,-21.54,;33.32,-20.2,;31.77,-20.2,;35.63,-21.54,;36.56,-22.83,;38,-22.31,;38,-20.77,;39.12,-19.74,;38.76,-18.25,;37.38,-17.78,;36.25,-18.82,;36.56,-20.3,;26.37,-24.22,;24.83,-24.22,;19.53,-22.78,;20.35,-21.49,;19.63,-20.1,;18.09,-20.05,;17.26,-21.34,;17.98,-22.67,)| Show InChI InChI=1S/C27H35N5OS/c1-30(23-7-3-2-4-8-23)27(33)28-22-13-11-21(12-14-22)15-16-31-17-19-32(20-18-31)26-24-9-5-6-10-25(24)34-29-26/h2-10,21-22H,11-20H2,1H3,(H,28,33)/t21-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207145

(CHEMBL3946995 | US9550741, I-1)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1ccco1 |r,wU:3.2,wD:6.6,(13.36,-22.55,;12.59,-21.22,;13.36,-19.89,;14.89,-19.89,;15.67,-18.56,;17.21,-18.56,;17.97,-19.89,;19.51,-19.89,;20.29,-18.56,;21.83,-18.56,;22.59,-17.22,;24.13,-17.22,;24.9,-18.56,;24.13,-19.89,;22.59,-19.89,;26.44,-18.56,;27.34,-19.8,;28.81,-19.32,;28.81,-17.78,;29.95,-16.75,;29.63,-15.25,;28.16,-14.78,;27.02,-15.81,;27.34,-17.31,;17.21,-21.22,;15.67,-21.22,;11.05,-21.22,;10.15,-19.97,;8.68,-20.45,;8.68,-21.98,;10.15,-22.46,)| Show InChI InChI=1S/C24H30N4O2S/c29-24(21-5-3-17-30-21)25-19-9-7-18(8-10-19)11-12-27-13-15-28(16-14-27)23-20-4-1-2-6-22(20)31-26-23/h1-6,17-19H,7-16H2,(H,25,29)/t18-,19- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50343260
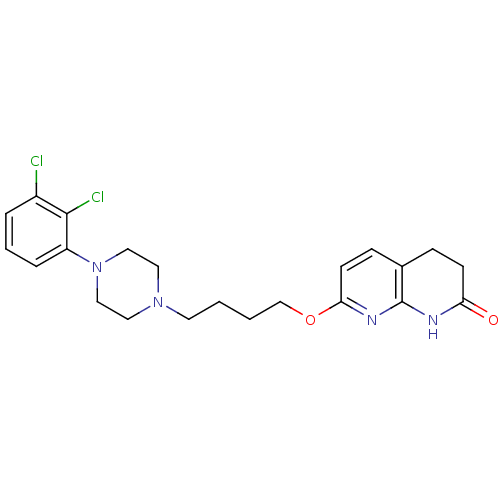
(7-(4-(4-(2,3-dichlorophenyl)piperazin-1-yl)butoxy)...)Show SMILES Clc1cccc(N2CCN(CCCCOc3ccc4CCC(=O)Nc4n3)CC2)c1Cl Show InChI InChI=1S/C22H26Cl2N4O2/c23-17-4-3-5-18(21(17)24)28-13-11-27(12-14-28)10-1-2-15-30-20-9-7-16-6-8-19(29)25-22(16)26-20/h3-5,7,9H,1-2,6,8,10-15H2,(H,25,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to human 5HT2A receptor |
Bioorg Med Chem Lett 21: 2621-5 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.059
BindingDB Entry DOI: 10.7270/Q29K4BJ3 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207091

(CHEMBL3953773 | US9550741, IV-6)Show SMILES CN(C(=O)N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1ccccc1 |r,wU:5.4,wD:8.8,(19.37,-25.45,;20.19,-24.17,;21.74,-24.22,;22.46,-25.6,;22.51,-22.88,;24.05,-22.88,;24.83,-21.54,;26.37,-21.54,;27.14,-22.88,;28.69,-22.88,;29.46,-21.54,;31,-21.54,;31.77,-22.88,;33.32,-22.88,;34.09,-21.54,;33.32,-20.2,;31.77,-20.2,;35.63,-21.54,;36.56,-22.83,;38,-22.31,;38,-20.77,;39.12,-19.74,;38.76,-18.25,;37.38,-17.78,;36.25,-18.82,;36.56,-20.3,;26.37,-24.22,;24.83,-24.22,;19.53,-22.78,;20.35,-21.49,;19.63,-20.1,;18.09,-20.05,;17.26,-21.34,;17.98,-22.67,)| Show InChI InChI=1S/C27H35N5OS/c1-30(23-7-3-2-4-8-23)27(33)28-22-13-11-21(12-14-22)15-16-31-17-19-32(20-18-31)26-24-9-5-6-10-25(24)34-29-26/h2-10,21-22H,11-20H2,1H3,(H,28,33)/t21-,22- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
Vitamin D3 receptor
(Homo sapiens (Human)) | BDBM50207145

(CHEMBL3946995 | US9550741, I-1)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1ccco1 |r,wU:3.2,wD:6.6,(13.36,-22.55,;12.59,-21.22,;13.36,-19.89,;14.89,-19.89,;15.67,-18.56,;17.21,-18.56,;17.97,-19.89,;19.51,-19.89,;20.29,-18.56,;21.83,-18.56,;22.59,-17.22,;24.13,-17.22,;24.9,-18.56,;24.13,-19.89,;22.59,-19.89,;26.44,-18.56,;27.34,-19.8,;28.81,-19.32,;28.81,-17.78,;29.95,-16.75,;29.63,-15.25,;28.16,-14.78,;27.02,-15.81,;27.34,-17.31,;17.21,-21.22,;15.67,-21.22,;11.05,-21.22,;10.15,-19.97,;8.68,-20.45,;8.68,-21.98,;10.15,-22.46,)| Show InChI InChI=1S/C24H30N4O2S/c29-24(21-5-3-17-30-21)25-19-9-7-18(8-10-19)11-12-27-13-15-28(16-14-27)23-20-4-1-2-6-22(20)31-26-23/h1-6,17-19H,7-16H2,(H,25,29)/t18-,19- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
The experiment is carried out by the method according to Journal of Pharmacology and Experimental Therapeutics 2010, 333(1): 328. With [3H]methyl-spi... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50343277
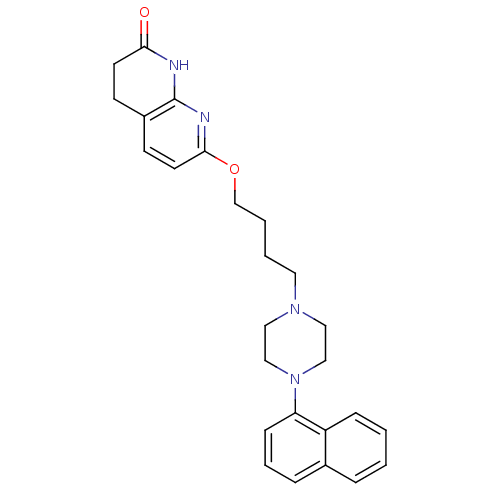
(7-(4-(4-(naphthalen-1-yl)piperazin-1-yl)butoxy)-3,...)Show SMILES O=C1CCc2ccc(OCCCCN3CCN(CC3)c3cccc4ccccc34)nc2N1 Show InChI InChI=1S/C26H30N4O2/c31-24-12-10-21-11-13-25(28-26(21)27-24)32-19-4-3-14-29-15-17-30(18-16-29)23-9-5-7-20-6-1-2-8-22(20)23/h1-2,5-9,11,13H,3-4,10,12,14-19H2,(H,27,28,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to human 5HT2A receptor |
Bioorg Med Chem Lett 21: 2621-5 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.059
BindingDB Entry DOI: 10.7270/Q29K4BJ3 |
More data for this
Ligand-Target Pair | |
Melanocortin receptor 4
(Homo sapiens (Human)) | BDBM50165929
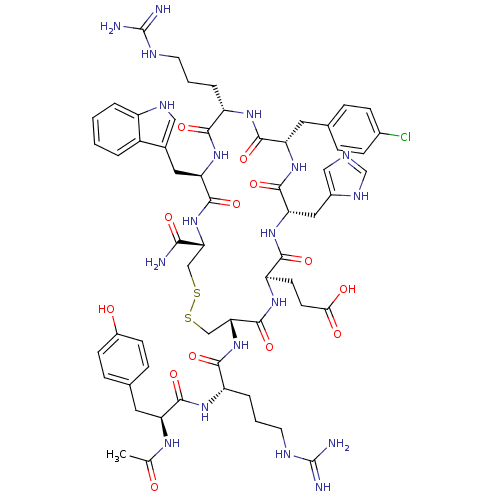
(Ac-YR[CEH(pCl-dF)RWC]-NH2 | CHEMBL415661)Show SMILES CC(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]1CSSC[C@@H](NC(=O)[C@@H](Cc2c[nH]c3ccccc23)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc2ccc(Cl)cc2)NC(=O)[C@H](Cc2cnc[nH]2)NC(=O)[C@@H](CCC(O)=O)NC1=O)C(N)=O Show InChI InChI=1S/C60H78ClN19O13S2/c1-31(81)72-43(22-33-12-16-37(82)17-13-33)54(89)73-41(9-5-21-69-60(65)66)52(87)80-48-29-95-94-28-47(50(62)85)79-56(91)45(24-34-26-70-39-7-3-2-6-38(34)39)77-51(86)40(8-4-20-68-59(63)64)74-55(90)44(23-32-10-14-35(61)15-11-32)76-57(92)46(25-36-27-67-30-71-36)78-53(88)42(75-58(48)93)18-19-49(83)84/h2-3,6-7,10-17,26-27,30,40-48,70,82H,4-5,8-9,18-25,28-29H2,1H3,(H2,62,85)(H,67,71)(H,72,81)(H,73,89)(H,74,90)(H,75,93)(H,76,92)(H,77,86)(H,78,88)(H,79,91)(H,80,87)(H,83,84)(H4,63,64,68)(H4,65,66,69)/t40-,41-,42+,43-,44-,45+,46-,47+,48+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of [125I]-NDP-alpha-MSH binding to melanocortin-4 receptor expressed in HEK293 cells |
J Med Chem 48: 3095-8 (2005)
Article DOI: 10.1021/jm0501432
BindingDB Entry DOI: 10.7270/Q2251HQZ |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50090003
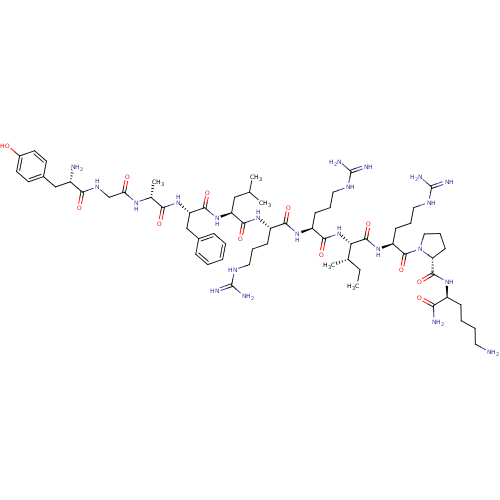
(CHEMBL405190 | Tyr-Gly-[D-Ala]-Phe-Leu-Arg-Arg-Ile...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](C)NC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCCN)C(N)=O Show InChI InChI=1S/C64H106N22O12/c1-6-37(4)51(60(97)82-46(21-14-30-76-64(72)73)61(98)86-31-15-22-49(86)59(96)79-43(52(67)89)18-10-11-27-65)85-56(93)45(20-13-29-75-63(70)71)80-55(92)44(19-12-28-74-62(68)69)81-57(94)47(32-36(2)3)84-58(95)48(34-39-16-8-7-9-17-39)83-53(90)38(5)78-50(88)35-77-54(91)42(66)33-40-23-25-41(87)26-24-40/h7-9,16-17,23-26,36-38,42-49,51,87H,6,10-15,18-22,27-35,65-66H2,1-5H3,(H2,67,89)(H,77,91)(H,78,88)(H,79,96)(H,80,92)(H,81,94)(H,82,97)(H,83,90)(H,84,95)(H,85,93)(H4,68,69,74)(H4,70,71,75)(H4,72,73,76)/t37-,38+,42-,43-,44-,45-,46-,47-,48-,49+,51-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
Human Opioid receptor kappa 1 mediated stimulation of [35S]- GTPgammaS binding in CHO cells (Agonist potency). |
J Med Chem 43: 2698-702 (2000)
BindingDB Entry DOI: 10.7270/Q25D8SJ1 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50207094

(CHEMBL3976282 | US9550741, I-2)Show SMILES O=C(N[C@H]1CC[C@H](CCN2CCN(CC2)c2nsc3ccccc23)CC1)c1cccs1 |r,wU:3.2,wD:6.6,(15.49,-22.15,;14.77,-20.77,;15.54,-19.44,;17.08,-19.44,;17.84,-18.11,;19.39,-18.11,;20.15,-19.44,;21.69,-19.44,;22.46,-18.11,;23.99,-18.11,;24.76,-19.44,;26.3,-19.44,;27.06,-18.11,;26.3,-16.77,;24.76,-16.77,;28.61,-18.11,;29.53,-19.33,;31.01,-18.87,;31.01,-17.34,;32.14,-16.31,;31.83,-14.78,;30.34,-14.31,;29.22,-15.34,;29.53,-16.88,;19.39,-20.77,;17.84,-20.77,;13.24,-20.67,;12.42,-19.38,;10.93,-19.79,;10.88,-21.34,;12.32,-21.9,)| Show InChI InChI=1S/C24H30N4OS2/c29-24(22-6-3-17-30-22)25-19-9-7-18(8-10-19)11-12-27-13-15-28(16-14-27)23-20-4-1-2-5-21(20)31-26-23/h1-6,17-19H,7-16H2,(H,25,29)/t18-,19- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | 7.7 | n/a |
Shanghai Institute of Pharmaceutical Industry
US Patent
| Assay Description
1. Experimental Materials(1) 5-HT2A Cell Transfection:This experiment utilizes the plasmid vector containing the gene of the 5-HT2A receptor protein ... |
US Patent US9550741 (2017)
BindingDB Entry DOI: 10.7270/Q2DN4726 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data


















































