| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50334937 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_702470 (CHEMBL1656984) |
|---|
| IC50 | 2000±n/a nM |
|---|
| Citation |  Aldegheri, L; Austin, NE; Ballantine, S; Bax, BD; Bradley, DM; Ballini, E; Harries, M; Harris, AJ; Clarke, BP; Mookherjee, C; Harrison, SA; Melarange, RA; Mosley, J; Oliosi, B; Dal Negro, G; Smith, KJ; Thewlis, KM; Ward, SE; Woollard, PM; Yusaf, SP Integration of lead optimization with crystallography for a membrane-bound ion channel target: discovery of a new class of AMPA receptor positive allosteric modulators. J Med Chem54:78-94 (2011) [PubMed] Article Aldegheri, L; Austin, NE; Ballantine, S; Bax, BD; Bradley, DM; Ballini, E; Harries, M; Harris, AJ; Clarke, BP; Mookherjee, C; Harrison, SA; Melarange, RA; Mosley, J; Oliosi, B; Dal Negro, G; Smith, KJ; Thewlis, KM; Ward, SE; Woollard, PM; Yusaf, SP Integration of lead optimization with crystallography for a membrane-bound ion channel target: discovery of a new class of AMPA receptor positive allosteric modulators. J Med Chem54:78-94 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50334937 |
|---|
| n/a |
|---|
| Name | BDBM50334937 |
|---|
| Synonyms: | 1-({2-Fluoro-4-[3-(trifluoromethyl)-4,5,6,7-tetrahydro-1Hindazol-1-yl]phenyl}methyl)-2-pyrrolidinone | CHEMBL1649674 | N,N-DIMETHYL-4-[3-(TRIFLUOROMETHYL)-4,5,6,7-TETRAHYDRO-1H-INDAZOL-1-YL]BENZAMIDE |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H19F4N3O |
|---|
| Mol. Mass. | 381.3673 |
|---|
| SMILES | Fc1cc(ccc1CN1CCCC1=O)-n1nc(c2CCCCc12)C(F)(F)F |
|---|
| Structure | 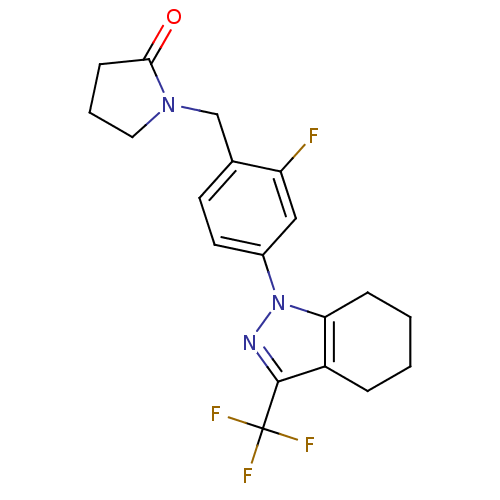
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Aldegheri, L; Austin, NE; Ballantine, S; Bax, BD; Bradley, DM; Ballini, E; Harries, M; Harris, AJ; Clarke, BP; Mookherjee, C; Harrison, SA; Melarange, RA; Mosley, J; Oliosi, B; Dal Negro, G; Smith, KJ; Thewlis, KM; Ward, SE; Woollard, PM; Yusaf, SP Integration of lead optimization with crystallography for a membrane-bound ion channel target: discovery of a new class of AMPA receptor positive allosteric modulators. J Med Chem54:78-94 (2011) [PubMed] Article
Aldegheri, L; Austin, NE; Ballantine, S; Bax, BD; Bradley, DM; Ballini, E; Harries, M; Harris, AJ; Clarke, BP; Mookherjee, C; Harrison, SA; Melarange, RA; Mosley, J; Oliosi, B; Dal Negro, G; Smith, KJ; Thewlis, KM; Ward, SE; Woollard, PM; Yusaf, SP Integration of lead optimization with crystallography for a membrane-bound ion channel target: discovery of a new class of AMPA receptor positive allosteric modulators. J Med Chem54:78-94 (2011) [PubMed] Article