| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50401097 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_884208 (CHEMBL2209438) |
|---|
| IC50 | 16000±n/a nM |
|---|
| Citation |  Kaila, N; Huang, A; Moretto, A; Follows, B; Janz, K; Lowe, M; Thomason, J; Mansour, TS; Hubeau, C; Page, K; Morgan, P; Fish, S; Xu, X; Williams, C; Saiah, E Diazine indole acetic acids as potent, selective, and orally bioavailable antagonists of chemoattractant receptor homologous molecule expressed on Th2 cells (CRTH2) for the treatment of allergic inflammatory diseases. J Med Chem55:5088-109 (2012) [PubMed] Article Kaila, N; Huang, A; Moretto, A; Follows, B; Janz, K; Lowe, M; Thomason, J; Mansour, TS; Hubeau, C; Page, K; Morgan, P; Fish, S; Xu, X; Williams, C; Saiah, E Diazine indole acetic acids as potent, selective, and orally bioavailable antagonists of chemoattractant receptor homologous molecule expressed on Th2 cells (CRTH2) for the treatment of allergic inflammatory diseases. J Med Chem55:5088-109 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50401097 |
|---|
| n/a |
|---|
| Name | BDBM50401097 |
|---|
| Synonyms: | CHEMBL2204469 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H18F3N3O3 |
|---|
| Mol. Mass. | 441.4025 |
|---|
| SMILES | Cc1c(Cc2ccc(=O)n(Cc3ccc(F)cc3F)n2)c2cc(F)ccc2n1CC(O)=O |
|---|
| Structure | 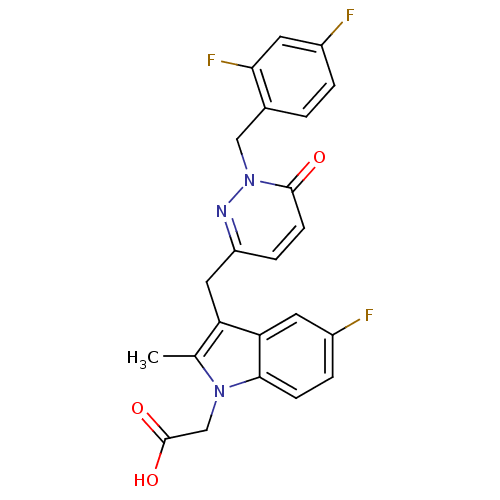
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kaila, N; Huang, A; Moretto, A; Follows, B; Janz, K; Lowe, M; Thomason, J; Mansour, TS; Hubeau, C; Page, K; Morgan, P; Fish, S; Xu, X; Williams, C; Saiah, E Diazine indole acetic acids as potent, selective, and orally bioavailable antagonists of chemoattractant receptor homologous molecule expressed on Th2 cells (CRTH2) for the treatment of allergic inflammatory diseases. J Med Chem55:5088-109 (2012) [PubMed] Article
Kaila, N; Huang, A; Moretto, A; Follows, B; Janz, K; Lowe, M; Thomason, J; Mansour, TS; Hubeau, C; Page, K; Morgan, P; Fish, S; Xu, X; Williams, C; Saiah, E Diazine indole acetic acids as potent, selective, and orally bioavailable antagonists of chemoattractant receptor homologous molecule expressed on Th2 cells (CRTH2) for the treatment of allergic inflammatory diseases. J Med Chem55:5088-109 (2012) [PubMed] Article