| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2B |
|---|
| Ligand | BDBM50403827 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_2888 |
|---|
| Ki | 631±n/a nM |
|---|
| Citation |  Bromidge, SM; Clarke, SE; Gager, T; Griffith, K; Jeffrey, P; Jennings, AJ; Joiner, GF; King, FD; Lovell, PJ; Moss, SF; Newman, H; Riley, G; Rogers, D; Routledge, C; Serafinowska, H; Smith, DR Phenyl benzenesulfonamides are novel and selective 5-HT6 antagonists: identification of N-(2,5-dibromo-3-fluorophenyl)-4-methoxy-3-piperazin-1-ylbenzenesulfonamide (SB-357134). Bioorg Med Chem Lett11:55-8 (2001) [PubMed] Bromidge, SM; Clarke, SE; Gager, T; Griffith, K; Jeffrey, P; Jennings, AJ; Joiner, GF; King, FD; Lovell, PJ; Moss, SF; Newman, H; Riley, G; Rogers, D; Routledge, C; Serafinowska, H; Smith, DR Phenyl benzenesulfonamides are novel and selective 5-HT6 antagonists: identification of N-(2,5-dibromo-3-fluorophenyl)-4-methoxy-3-piperazin-1-ylbenzenesulfonamide (SB-357134). Bioorg Med Chem Lett11:55-8 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2B |
|---|
| Name: | 5-hydroxytryptamine receptor 2B |
|---|
| Synonyms: | 5-HT-2B | 5-HT2B | 5-hydroxytryptamine (serotonin) receptor 2B [Homo sapiens] | 5-hydroxytryptamine receptor 2B (5-HT2B) | 5-hydroxytryptamine receptor 2C (5HT2C) | 5HT2B_HUMAN | HTR2B | Serotonin (5-HT3) receptor | Serotonin 2b (5-HT2b) receptor | Serotonin Receptor 2B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 54312.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Receptor binding assays were performed using human clone stably expressed in CHO cells. |
|---|
| Residue: | 481 |
|---|
| Sequence: | MALSYRVSELQSTIPEHILQSTFVHVISSNWSGLQTESIPEEMKQIVEEQGNKLHWAALL
ILMVIIPTIGGNTLVILAVSLEKKLQYATNYFLMSLAVADLLVGLFVMPIALLTIMFEAM
WPLPLVLCPAWLFLDVLFSTASIMHLCAISVDRYIAIKKPIQANQYNSRATAFIKITVVW
LISIGIAIPVPIKGIETDVDNPNNITCVLTKERFGDFMLFGSLAAFFTPLAIMIVTYFLT
IHALQKKAYLVKNKPPQRLTWLTVSTVFQRDETPCSSPEKVAMLDGSRKDKALPNSGDET
LMRRTSTIGKKSVQTISNEQRASKVLGIVFFLFLLMWCPFFITNITLVLCDSCNQTTLQM
LLEIFVWIGYVSSGVNPLVYTLFNKTFRDAFGRYITCNYRATKSVKTLRKRSSKIYFRNP
MAENSKFFKKHGIRNGINPAMYQSPMRLRSSTIQSSSIILLDTLLLTENEGDKTEEQVSY
V
|
|
|
|---|
| BDBM50403827 |
|---|
| n/a |
|---|
| Name | BDBM50403827 |
|---|
| Synonyms: | CHEMBL414628 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H22IN3O3S |
|---|
| Mol. Mass. | 499.366 |
|---|
| SMILES | COc1ccc(cc1N1CCNCC1)S(=O)(=O)N1CCc2ccc(I)cc12 |
|---|
| Structure | 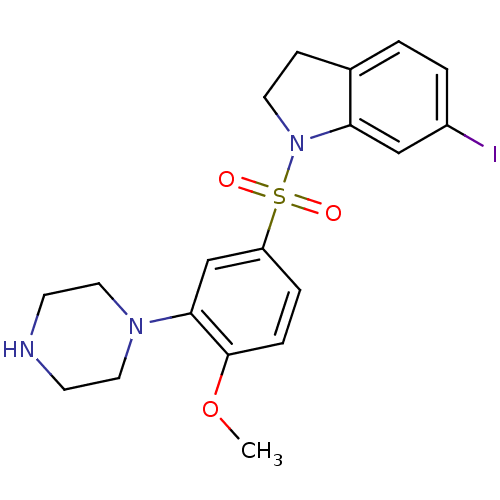
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bromidge, SM; Clarke, SE; Gager, T; Griffith, K; Jeffrey, P; Jennings, AJ; Joiner, GF; King, FD; Lovell, PJ; Moss, SF; Newman, H; Riley, G; Rogers, D; Routledge, C; Serafinowska, H; Smith, DR Phenyl benzenesulfonamides are novel and selective 5-HT6 antagonists: identification of N-(2,5-dibromo-3-fluorophenyl)-4-methoxy-3-piperazin-1-ylbenzenesulfonamide (SB-357134). Bioorg Med Chem Lett11:55-8 (2001) [PubMed]
Bromidge, SM; Clarke, SE; Gager, T; Griffith, K; Jeffrey, P; Jennings, AJ; Joiner, GF; King, FD; Lovell, PJ; Moss, SF; Newman, H; Riley, G; Rogers, D; Routledge, C; Serafinowska, H; Smith, DR Phenyl benzenesulfonamides are novel and selective 5-HT6 antagonists: identification of N-(2,5-dibromo-3-fluorophenyl)-4-methoxy-3-piperazin-1-ylbenzenesulfonamide (SB-357134). Bioorg Med Chem Lett11:55-8 (2001) [PubMed]