| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50135154 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_29848 |
|---|
| IC50 | 181970±n/a nM |
|---|
| Citation |  Zaheer-ul-Haq, na; Wellenzohn, B; Tonmunphean, S; Khalid, A; Choudhary, MI; Rode, BM 3D-QSAR studies on natural acetylcholinesterase inhibitors of Sarcococca saligna by comparative molecular field analysis (CoMFA). Bioorg Med Chem Lett13:4375-80 (2003) [PubMed] Zaheer-ul-Haq, na; Wellenzohn, B; Tonmunphean, S; Khalid, A; Choudhary, MI; Rode, BM 3D-QSAR studies on natural acetylcholinesterase inhibitors of Sarcococca saligna by comparative molecular field analysis (CoMFA). Bioorg Med Chem Lett13:4375-80 (2003) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_HUMAN | ACHE | Acetylcholinesterase (AChE) | Acetylcholinesterase (human AChE) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67792.70 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22303 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPV
SAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSM
NYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASV
GMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTEL
VACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVG
VVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPE
DPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGY
EIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQ
YVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQDRCSDL
|
|
|
|---|
| BDBM50135154 |
|---|
| n/a |
|---|
| Name | BDBM50135154 |
|---|
| Synonyms: | Acetic acid (1S,4S,6R,7R,10R,11S,12S,15S,16S)-17-((S)-1-dimethylamino-ethyl)-2-(S)-hydroxy-10,13-dimethyl-3-((E)-2-methyl-but-2-enoylamino)-hexadecahydro-cyclopenta[a]phenanthren-4-yl ester | Acetic acid (2S,3S,4R,5R,8S,9S,10R,13S,14S,17S)-17-((S)-1-dimethylamino-ethyl)-2-hydroxy-10,13-dimethyl-3-((E)-2-methyl-but-2-enoylamino)-hexadecahydro-cyclopenta[a]phenanthren-4-yl ester | CHEMBL421920 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H50N2O4 |
|---|
| Mol. Mass. | 502.729 |
|---|
| SMILES | C\C=C(/C)C(=O)N[C@H]1[C@@H](O)C[C@]2(C)[C@H]3CC[C@]4(C)[C@H](CC[C@H]4[C@@H]3CC[C@H]2[C@H]1OC(C)=O)[C@H](C)N(C)C |
|---|
| Structure | 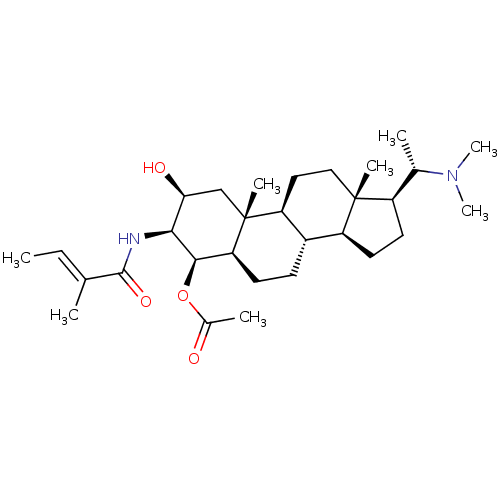
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zaheer-ul-Haq, na; Wellenzohn, B; Tonmunphean, S; Khalid, A; Choudhary, MI; Rode, BM 3D-QSAR studies on natural acetylcholinesterase inhibitors of Sarcococca saligna by comparative molecular field analysis (CoMFA). Bioorg Med Chem Lett13:4375-80 (2003) [PubMed]
Zaheer-ul-Haq, na; Wellenzohn, B; Tonmunphean, S; Khalid, A; Choudhary, MI; Rode, BM 3D-QSAR studies on natural acetylcholinesterase inhibitors of Sarcococca saligna by comparative molecular field analysis (CoMFA). Bioorg Med Chem Lett13:4375-80 (2003) [PubMed]