| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Serine/threonine-protein kinase TBK1 |
|---|
| Ligand | BDBM27452 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1297370 (CHEMBL3132510) |
|---|
| IC50 | 72±n/a nM |
|---|
| Citation |  Johannes, JW; Chuaqui, C; Cowen, S; Devereaux, E; Gingipalli, L; Molina, A; Wang, T; Whitston, D; Wu, X; Zhang, HJ; Zinda, M Discovery of 6-aryl-azabenzimidaoles that inhibit the TBK1/IKK-e kinases. Bioorg Med Chem Lett24:1138-43 (2014) [PubMed] Article Johannes, JW; Chuaqui, C; Cowen, S; Devereaux, E; Gingipalli, L; Molina, A; Wang, T; Whitston, D; Wu, X; Zhang, HJ; Zinda, M Discovery of 6-aryl-azabenzimidaoles that inhibit the TBK1/IKK-e kinases. Bioorg Med Chem Lett24:1138-43 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Serine/threonine-protein kinase TBK1 |
|---|
| Name: | Serine/threonine-protein kinase TBK1 |
|---|
| Synonyms: | NAK | NF-kappa-B-activating kinase | Protein cereblon/Serine/threonine-protein kinase TBK1 | T2K | TANK-binding kinase 1 (TBK-1) | TANK-binding kinase 1 (TBK1) | TBK1 | TBK1_HUMAN |
|---|
| Type: | protein |
|---|
| Mol. Mass.: | 83645.20 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q9UHD2 |
|---|
| Residue: | 729 |
|---|
| Sequence: | MQSTSNHLWLLSDILGQGATANVFRGRHKKTGDLFAIKVFNNISFLRPVDVQMREFEVLK
KLNHKNIVKLFAIEEETTTRHKVLIMEFCPCGSLYTVLEEPSNAYGLPESEFLIVLRDVV
GGMNHLRENGIVHRDIKPGNIMRVIGEDGQSVYKLTDFGAARELEDDEQFVSLYGTEEYL
HPDMYERAVLRKDHQKKYGATVDLWSIGVTFYHAATGSLPFRPFEGPRRNKEVMYKIITG
KPSGAISGVQKAENGPIDWSGDMPVSCSLSRGLQVLLTPVLANILEADQEKCWGFDQFFA
ETSDILHRMVIHVFSLQQMTAHKIYIHSYNTATIFHELVYKQTKIISSNQELIYEGRRLV
LEPGRLAQHFPKTTEENPIFVVSREPLNTIGLIYEKISLPKVHPRYDLDGDASMAKAITG
VVCYACRIASTLLLYQELMRKGIRWLIELIKDDYNETVHKKTEVVITLDFCIRNIEKTVK
VYEKLMKINLEAAELGEISDIHTKLLRLSSSQGTIETSLQDIDSRLSPGGSLADAWAHQE
GTHPKDRNVEKLQVLLNCMTEIYYQFKKDKAERRLAYNEEQIHKFDKQKLYYHATKAMTH
FTDECVKKYEAFLNKSEEWIRKMLHLRKQLLSLTNQCFDIEEEVSKYQEYTNELQETLPQ
KMFTASSGIKHTMTPIYPSSNTLVEMTLGMKKLKEEMEGVVKELAENNHILERFGSLTMD
GGLRNVDCL
|
|
|
|---|
| BDBM27452 |
|---|
| n/a |
|---|
| Name | BDBM27452 |
|---|
| Synonyms: | 5-(5,6-dimethoxy-1H-1,3-benzodiazol-1-yl)-3-[(2-methanesulfonylphenyl)methoxy]thiophene-2-carbonitrile | benzimidazole-thiophene carbonitrile, 12e |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H19N3O5S2 |
|---|
| Mol. Mass. | 469.533 |
|---|
| SMILES | COc1cc2ncn(-c3cc(OCc4ccccc4S(C)(=O)=O)c(s3)C#N)c2cc1OC |
|---|
| Structure | 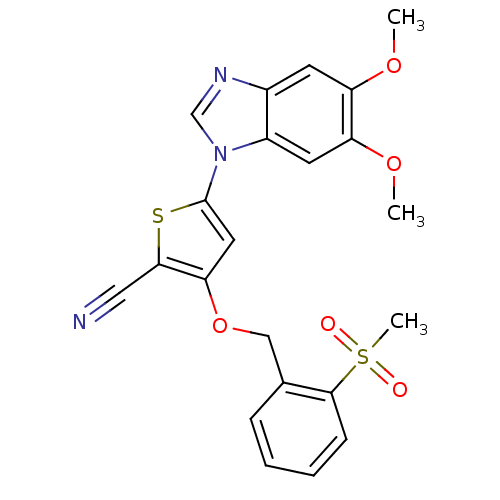
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Johannes, JW; Chuaqui, C; Cowen, S; Devereaux, E; Gingipalli, L; Molina, A; Wang, T; Whitston, D; Wu, X; Zhang, HJ; Zinda, M Discovery of 6-aryl-azabenzimidaoles that inhibit the TBK1/IKK-e kinases. Bioorg Med Chem Lett24:1138-43 (2014) [PubMed] Article
Johannes, JW; Chuaqui, C; Cowen, S; Devereaux, E; Gingipalli, L; Molina, A; Wang, T; Whitston, D; Wu, X; Zhang, HJ; Zinda, M Discovery of 6-aryl-azabenzimidaoles that inhibit the TBK1/IKK-e kinases. Bioorg Med Chem Lett24:1138-43 (2014) [PubMed] Article