| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholinesterase |
|---|
| Ligand | BDBM50210773 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1635127 (CHEMBL3878025) |
|---|
| Ki | 7510±n/a nM |
|---|
| Citation |  Loesche, A; Wiese, J; Sommerwerk, S; Simon, V; Brandt, W; Csuk, R Repurposing N,N'-bis-(arylamidino)-1,4-piperazinedicarboxamidines: An unexpected class of potent inhibitors of cholinesterases. Eur J Med Chem125:430-434 (2017) [PubMed] Article Loesche, A; Wiese, J; Sommerwerk, S; Simon, V; Brandt, W; Csuk, R Repurposing N,N'-bis-(arylamidino)-1,4-piperazinedicarboxamidines: An unexpected class of potent inhibitors of cholinesterases. Eur J Med Chem125:430-434 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholinesterase |
|---|
| Name: | Cholinesterase |
|---|
| Synonyms: | BCHE | Butyrylcholinesterase (BuChE) | CHLE_HORSE | Cholinesterase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 65643.35 |
|---|
| Organism: | Equus caballus (Horse) |
|---|
| Description: | P81908 |
|---|
| Residue: | 574 |
|---|
| Sequence: | EEDIIITTKNGKVRGMNLPVLGGTVTAFLGIPYAQPPLGRLRFKKPQSLTKWSNIWNATK
YANSCYQNTDQSFPGFLGSEMWNPNTELSEDCLYLNVWIPAPKPKNATVMIWIYGGGFQT
GTSSLPVYDGKFLARVERVIVVSMNYRVGALGFLALSENPEAPGNMGLFDQQLALQWVQK
NIAAFGGNPRSVTLFGESAGAASVSLHLLSPRSQPLFTRAILQSGSSNAPWAVTSLYEAR
NRTLTLAKRMGCSRDNETEMIKCLRDKDPQEILLNEVFVVPYDTLLSVNFGPTVDGDFLT
DMPDTLLQLGQFKRTQILVGVNKDEGTAFLVYGAPGFSKDNNSIITRKEFQEGLKIFFPR
VSEFGRESILFHYMDWLDDQRAENYREALDDVVGDYNIICPALEFTRKFSELGNDAFFYY
FEHRSTKLPWPEWMGVMHGYEIEFVFGLPLERRVNYTRAEEILSRSIMKRWANFAKYGNP
NGTQNNSTRWPVFKSTEQKYLTLNTESPKVYTKLRAQQCRFWTLFFPKVLELTGNIDEAE
REWKAGFHRWNNYMMDWKNQFNDYTSKKESCSDF
|
|
|
|---|
| BDBM50210773 |
|---|
| n/a |
|---|
| Name | BDBM50210773 |
|---|
| Synonyms: | CHEMBL3968731 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H22Cl4N10 |
|---|
| Mol. Mass. | 544.268 |
|---|
| SMILES | Clc1ccc(NC(=N)NC(=N)N2CCN(CC2)C(=N)NC(=N)Nc2ccc(Cl)cc2Cl)c(Cl)c1 |
|---|
| Structure | 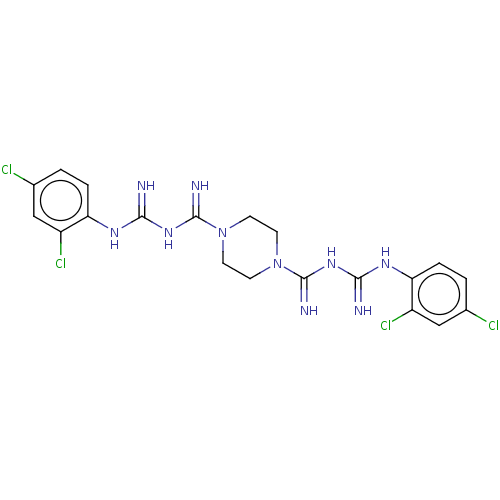
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Loesche, A; Wiese, J; Sommerwerk, S; Simon, V; Brandt, W; Csuk, R Repurposing N,N'-bis-(arylamidino)-1,4-piperazinedicarboxamidines: An unexpected class of potent inhibitors of cholinesterases. Eur J Med Chem125:430-434 (2017) [PubMed] Article
Loesche, A; Wiese, J; Sommerwerk, S; Simon, V; Brandt, W; Csuk, R Repurposing N,N'-bis-(arylamidino)-1,4-piperazinedicarboxamidines: An unexpected class of potent inhibitors of cholinesterases. Eur J Med Chem125:430-434 (2017) [PubMed] Article