

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Amine oxidase [flavin-containing] B | ||
| Ligand | BDBM254555 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | Fluorescence-Based (Inhibitor)-Screening Assay | ||
| pH | 7.5±n/a | ||
| Temperature | 310.15±n/a K | ||
| Ki | 20000±n/a nM | ||
| Comments | extracted | ||
| Citation |  Ortega Muñoz, A; Fyfe, MC; Martinell Pedemonte, M; Estiarte Martínez, M; Valls Vidal, N; Kurz, G; Castro Palomino Laria, JC (Hetero)aryl cyclopropylamine compounds as LSD1 inhibitors US Patent US9469597 Publication Date 10/18/2016 Ortega Muñoz, A; Fyfe, MC; Martinell Pedemonte, M; Estiarte Martínez, M; Valls Vidal, N; Kurz, G; Castro Palomino Laria, JC (Hetero)aryl cyclopropylamine compounds as LSD1 inhibitors US Patent US9469597 Publication Date 10/18/2016 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Amine oxidase [flavin-containing] B | |||
| Name: | Amine oxidase [flavin-containing] B | ||
| Synonyms: | AOFB_HUMAN | MAO-B | MAOB | Monoamine oxidase type B | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAOB) | ||
| Type: | Protein | ||
| Mol. Mass.: | 58768.76 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P27338 | ||
| Residue: | 520 | ||
| Sequence: |
| ||
| BDBM254555 | |||
| n/a | |||
| Name | BDBM254555 | ||
| Synonyms: | US10214477, Example 14 | US9469597, 14 | US9670136, 14 N-(4-(((trans)-2-(6-(3-(trifluoromethyl)phenyl)pyridin-3-yl)cyclopropyl)amino)cyclohexyl)methanesulfonamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C22H26F3N3O2S | ||
| Mol. Mass. | 453.521 | ||
| SMILES | CS(=O)(=O)NC1CCC(CC1)N[C@H]1C[C@@H]1c1ccc(nc1)-c1cccc(c1)C(F)(F)F |r,wU:14.16,wD:12.12,(9.54,3.69,;8.21,4.46,;9.3,5.55,;7.44,3.13,;6.87,5.23,;5.54,4.46,;5.54,2.92,;4.21,2.15,;2.87,2.92,;2.87,4.46,;4.21,5.23,;1.54,2.15,;.21,2.92,;-.56,4.25,;-1.33,2.92,;-2.67,2.15,;-4,2.92,;-5.33,2.15,;-5.33,.61,;-4,-.16,;-2.67,.61,;-6.67,-.16,;-8,.61,;-9.34,-.16,;-9.34,-1.7,;-8,-2.52,;-6.67,-1.7,;-8,-4.06,;-8,-5.6,;-9.54,-4.06,;-6.46,-4.06,)| | ||
| Structure | 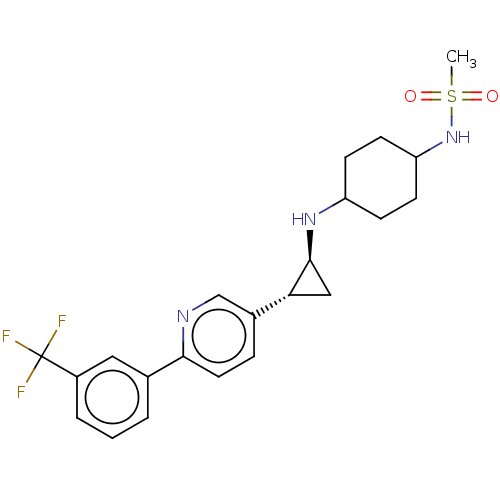
| ||