

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254546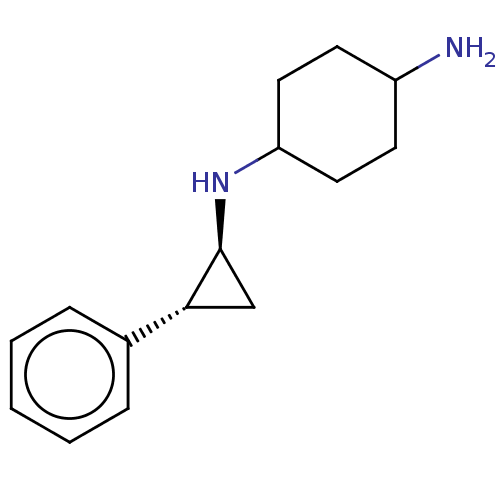 (US10214477, Example 3 | US9469597, 1 | US9670136, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US10214477 (2019) BindingDB Entry DOI: 10.7270/Q2ZS2ZTB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876] (Homo sapiens (Human)) | BDBM256461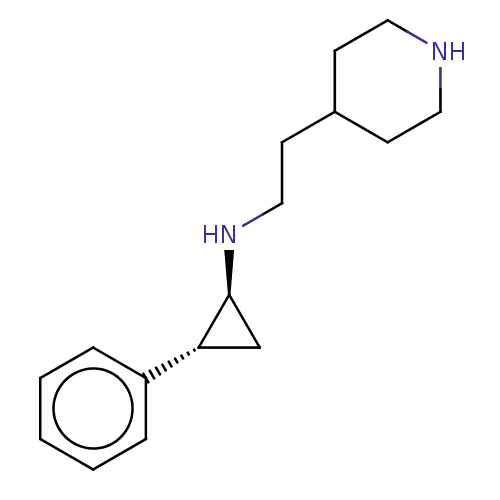 (US10329256, Example 6 | US9487512, 6 | US9944601, ...) | PDB UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9487512 (2016) BindingDB Entry DOI: 10.7270/Q2765D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876] (Homo sapiens (Human)) | BDBM256460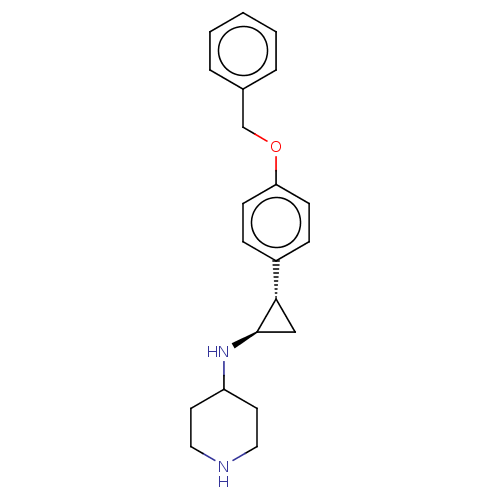 (US9487512, 4) | PDB UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9487512 (2016) BindingDB Entry DOI: 10.7270/Q2765D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876] (Homo sapiens (Human)) | BDBM256459 (US10329256, Example 3 | US9487512, 3 | US9944601, ...) | PDB UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9487512 (2016) BindingDB Entry DOI: 10.7270/Q2765D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876] (Homo sapiens (Human)) | BDBM256457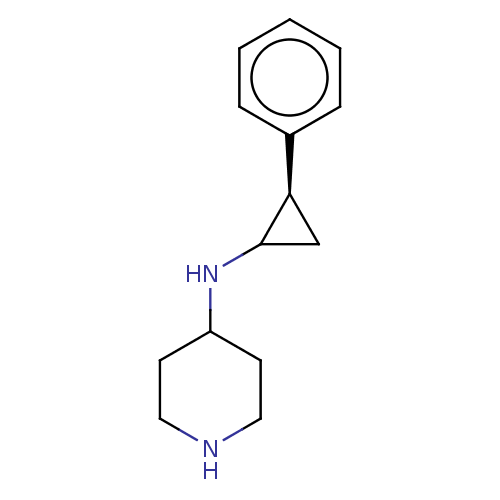 (US9487512, 1) | PDB UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9487512 (2016) BindingDB Entry DOI: 10.7270/Q2765D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254556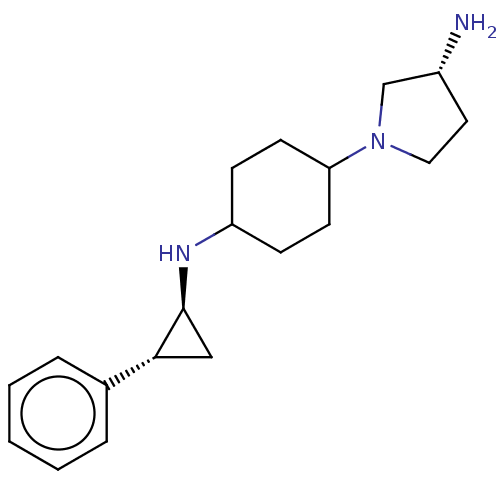 (US10214477, Example 15 | US9469597, 15 | US9670136...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254553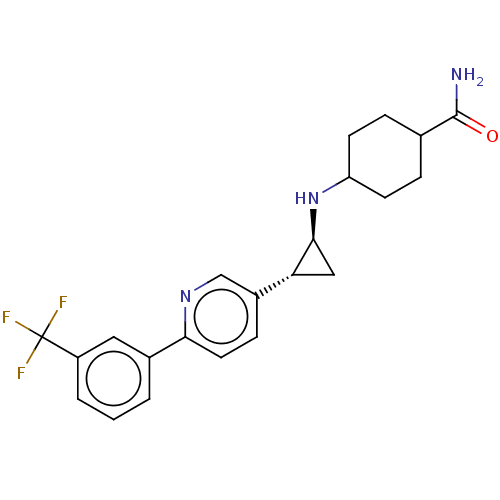 (US9469597, 12 | US9670136, 12 4-(((trans)-2-(6-(3-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM50155773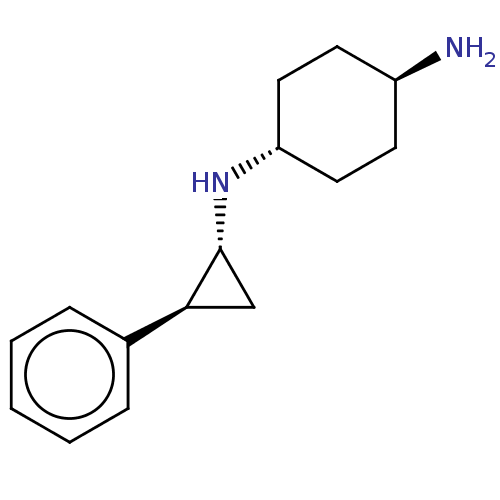 (CHEMBL3781751 | US9469597, 5) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254603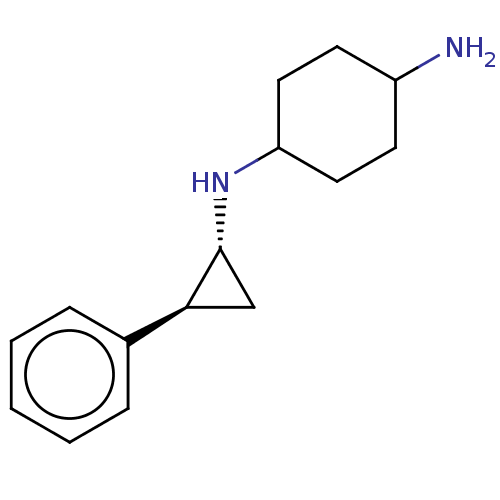 (US10214477, Example 5 | US9469597, 4 | US9670136, ...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254546 (US10214477, Example 3 | US9469597, 1 | US9670136, ...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876] (Homo sapiens (Human)) | BDBM50142189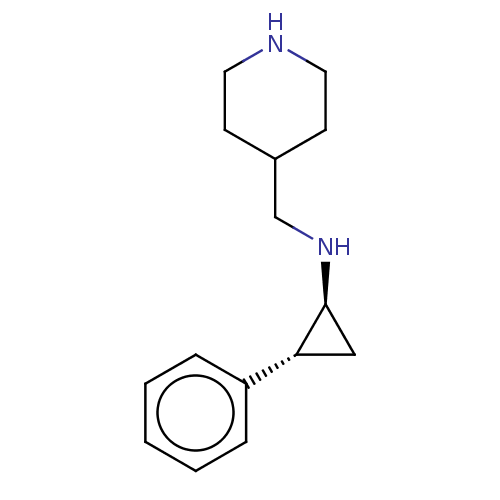 (CHEMBL3759239 | US10329256, Example 5 | US9487512,...) | PDB UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | US Patent | 50 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9487512 (2016) BindingDB Entry DOI: 10.7270/Q2765D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254546 (US10214477, Example 3 | US9469597, 1 | US9670136, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 50 | -41.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254546 (US10214477, Example 3 | US9469597, 1 | US9670136, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 50 | -41.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254553 (US9469597, 12 | US9670136, 12 4-(((trans)-2-(6-(3-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 50 | -41.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254556 (US10214477, Example 15 | US9469597, 15 | US9670136...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 50 | -41.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM182862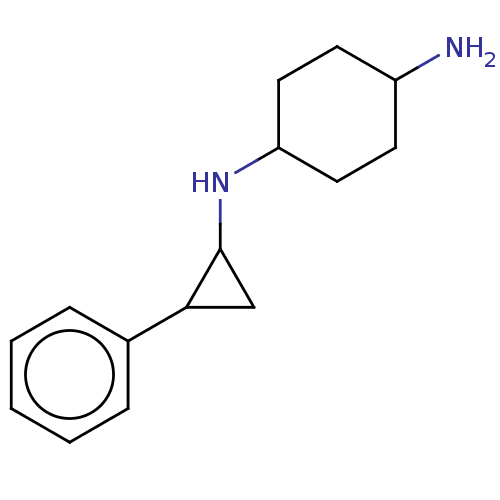 (US9670136, 1 N1-((trans)-2-phenylcyclopropyl)cyclo...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 50 | -41.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254603 (US10214477, Example 5 | US9469597, 4 | US9670136, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | US Patent | 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US10214477 (2019) BindingDB Entry DOI: 10.7270/Q2ZS2ZTB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254603 (US10214477, Example 5 | US9469597, 4 | US9670136, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | US Patent | 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US10214477 (2019) BindingDB Entry DOI: 10.7270/Q2ZS2ZTB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM356626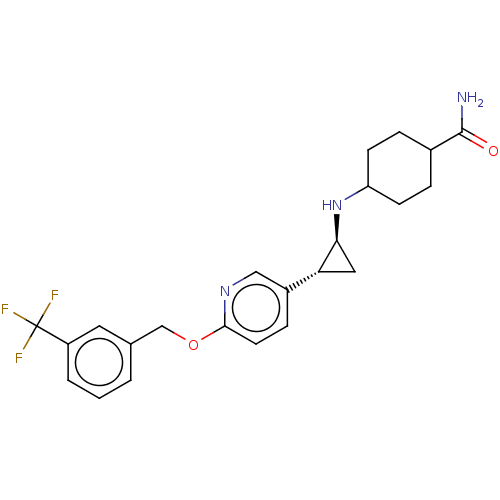 (US10214477, Example 12) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US10214477 (2019) BindingDB Entry DOI: 10.7270/Q2ZS2ZTB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254556 (US10214477, Example 15 | US9469597, 15 | US9670136...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oryzon Genomics S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US10214477 (2019) BindingDB Entry DOI: 10.7270/Q2ZS2ZTB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256458 (US10329256, Example 2 | US9487512, 2 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | Bioorg Med Chem Lett 19: 1164-7 (2009) BindingDB Entry DOI: 10.7270/Q2FX7CTX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256461 (US10329256, Example 6 | US9487512, 6 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | Bioorg Med Chem 17: 7324-36 (2009) BindingDB Entry DOI: 10.7270/Q2RF5XBG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50142189 (CHEMBL3759239 | US10329256, Example 5 | US9487512,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | Bioorg Med Chem 17: 7324-36 (2009) BindingDB Entry DOI: 10.7270/Q2RF5XBG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM388464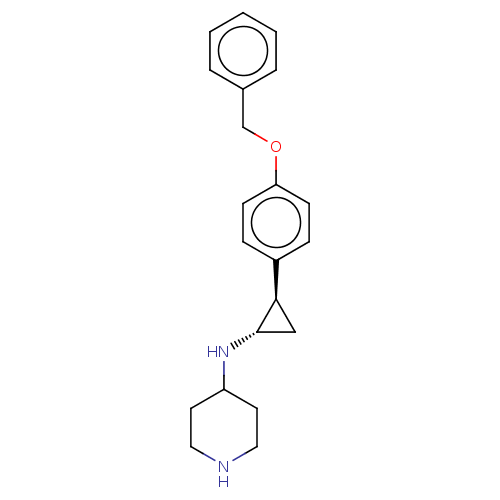 (US10329256, Example 4 | US9944601, Example 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | Bioorg Med Chem 17: 7324-36 (2009) BindingDB Entry DOI: 10.7270/Q2RF5XBG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256459 (US10329256, Example 3 | US9487512, 3 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | Bioorg Med Chem Lett 19: 1164-7 (2009) BindingDB Entry DOI: 10.7270/Q2FX7CTX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256458 (US10329256, Example 2 | US9487512, 2 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | Bioorg Med Chem 17: 7324-36 (2009) BindingDB Entry DOI: 10.7270/Q2RF5XBG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256461 (US10329256, Example 6 | US9487512, 6 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | Bioorg Med Chem Lett 19: 1164-7 (2009) BindingDB Entry DOI: 10.7270/Q2FX7CTX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50142189 (CHEMBL3759239 | US10329256, Example 5 | US9487512,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | Bioorg Med Chem Lett 19: 1164-7 (2009) BindingDB Entry DOI: 10.7270/Q2FX7CTX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM388464 (US10329256, Example 4 | US9944601, Example 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | Bioorg Med Chem Lett 19: 1164-7 (2009) BindingDB Entry DOI: 10.7270/Q2FX7CTX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256459 (US10329256, Example 3 | US9487512, 3 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | US Patent | 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | Bioorg Med Chem 17: 7324-36 (2009) BindingDB Entry DOI: 10.7270/Q2RF5XBG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256462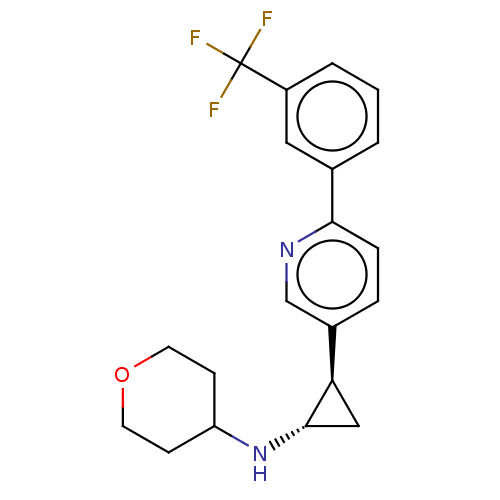 (US10329256, Example 7 | US9487512, 7 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | Bioorg Med Chem 17: 7324-36 (2009) BindingDB Entry DOI: 10.7270/Q2RF5XBG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254601 (US9469597, 2) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 550 | -37.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876] (Homo sapiens (Human)) | BDBM256458 (US10329256, Example 2 | US9487512, 2 | US9944601, ...) | PDB UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 550 | -37.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9487512 (2016) BindingDB Entry DOI: 10.7270/Q2765D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM182862 (US9670136, 1 N1-((trans)-2-phenylcyclopropyl)cyclo...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 550 | -35.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256462 (US10329256, Example 7 | US9487512, 7 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | Bioorg Med Chem Lett 19: 1164-7 (2009) BindingDB Entry DOI: 10.7270/Q2FX7CTX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256458 (US10329256, Example 2 | US9487512, 2 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | Bioorg Med Chem 17: 7324-36 (2009) BindingDB Entry DOI: 10.7270/Q2RF5XBG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254555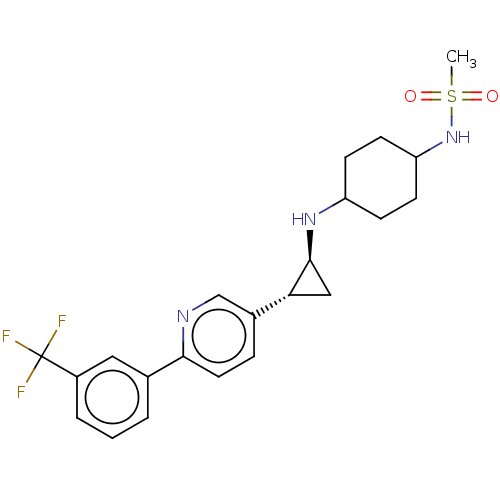 (US10214477, Example 14 | US9469597, 14 | US9670136...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | -37.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254554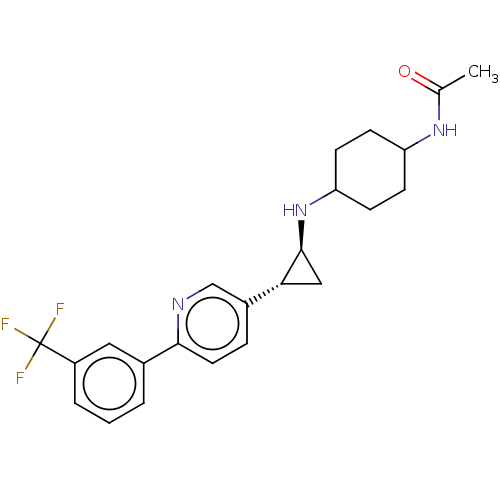 (US10214477, Example 13 | US9469597, 13 | US9670136...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | -37.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254552 (US9469597, 11 | US9670136, 11 4-(((trans)-2-(6-(3-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | -37.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254549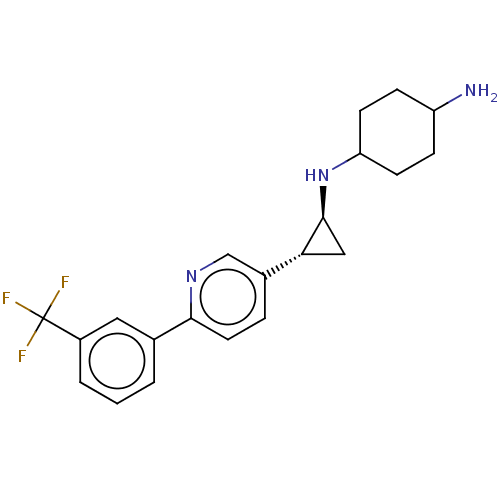 (US10214477, Example 8 | US9469597, 8 | US9670136, ...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | -37.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Lysine-specific histone demethylase 1A [158-852] (Homo sapiens (Human)) | BDBM254602 (US9469597, 3) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 550 | -37.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9469597 (2016) BindingDB Entry DOI: 10.7270/Q2ZW1JVG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256458 (US10329256, Example 2 | US9487512, 2 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | US Patent | 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | Bioorg Med Chem Lett 19: 1164-7 (2009) BindingDB Entry DOI: 10.7270/Q2FX7CTX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256464 (US10329256, Example 9 | US9487512, 9 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | Bioorg Med Chem Lett 19: 1164-7 (2009) BindingDB Entry DOI: 10.7270/Q2FX7CTX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM256464 (US10329256, Example 9 | US9487512, 9 | US9944601, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Duquesne University | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | Bioorg Med Chem 17: 7324-36 (2009) BindingDB Entry DOI: 10.7270/Q2RF5XBG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glutathione S-transferase P/Isoform 2 of Lysine-specific histone demethylase 1A (2) [158-876] (Homo sapiens (Human)) | BDBM256464 (US10329256, Example 9 | US9487512, 9 | US9944601, ...) | PDB UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | -37.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Oryzon Genomics S.A. US Patent | Assay Description The compounds of the invention can be tested for their ability to inhibit LSD1. The ability of the compounds of the invention to inhibit LSD1 can be ... | US Patent US9487512 (2016) BindingDB Entry DOI: 10.7270/Q2765D89 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254603 (US10214477, Example 5 | US9469597, 4 | US9670136, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | US Patent | 550 | -35.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254549 (US10214477, Example 8 | US9469597, 8 | US9670136, ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | -35.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254552 (US9469597, 11 | US9670136, 11 4-(((trans)-2-(6-(3-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | -35.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254554 (US10214477, Example 13 | US9469597, 13 | US9670136...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | -35.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM254555 (US10214477, Example 14 | US9469597, 14 | US9670136...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 550 | -35.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
ORYZON GENOMICS S.A. US Patent | Assay Description Briefly, a fixed amount of LSD1 was incubated on ice for 15 minutes, in the absence and/or in the presence of at least eight 3-fold serial dilutions ... | US Patent US9670136 (2017) BindingDB Entry DOI: 10.7270/Q2PN93SB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 1188 total ) | Next | Last >> |