| Reaction Details |
|---|
 | Report a problem with these data |
| Target | DNA dC->dU-editing enzyme APOBEC-3G |
|---|
| Ligand | BDBM38617 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Dose Response confirmation of small molecule APOBEC3G DNA Deaminase Inhibitors via a fluorescence-based single-stranded DNA deaminase assay |
|---|
| Temperature | 298.15±n/a K |
|---|
| IC50 | 2500±n/a nM |
|---|
| Comments | extracted |
|---|
| Citation |  PubChem, PC Dose Response confirmation of small molecule APOBEC3G DNA Deaminase Inhibitors via a fluorescence-based single-stranded DNA deaminase assay PubChem Bioassay(2011)[AID] PubChem, PC Dose Response confirmation of small molecule APOBEC3G DNA Deaminase Inhibitors via a fluorescence-based single-stranded DNA deaminase assay PubChem Bioassay(2011)[AID] |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| DNA dC->dU-editing enzyme APOBEC-3G |
|---|
| Name: | DNA dC->dU-editing enzyme APOBEC-3G |
|---|
| Synonyms: | ABC3G_HUMAN | APOBEC3G |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 46419.54 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_13399304 |
|---|
| Residue: | 384 |
|---|
| Sequence: | MKPHFRNTVERMYRDTFSYNFYNRPILSRRNTVWLCYEVKTKGPSRPPLDAKIFRGQVYS
ELKYHPEMRFFHWFSKWRKLHRDQEYEVTWYISWSPCTKCTRDMATFLAEDPKVTLTIFV
ARLYYFWDPDYQEALRSLCQKRDGPRATMKIMNYDEFQHCWSKFVYSQRELFEPWNNLPK
YYILLHIMLGEILRHSMDPPTFTFNFNNEPWVRGRHETYLCYEVERMHNDTWVLLNQRRG
FLCNQAPHKHGFLEGRHAELCFLDVIPFWKLDLDQDYRVTCFTSWSPCFSCAQEMAKFIS
KNKHVSLCIFTARIYDDQGRCQEGLRTLAEAGAKISIMTYSEFKHCWDTFVDHQGCPFQP
WDGLDEHSQDLSGRLRAILQNQEN
|
|
|
|---|
| BDBM38617 |
|---|
| n/a |
|---|
| Name | BDBM38617 |
|---|
| Synonyms: | MLS000092916 | N-[2-(diethylamino)ethyl]-5-(4-keto-8-methoxy-2-thioxo-1,5-dihydropyrimid[5,4-b]indol-3-yl)valeramide | N-[2-(diethylamino)ethyl]-5-(8-methoxy-4-oxidanylidene-2-sulfanylidene-1,5-dihydropyrimido[5,4-b]indol-3-yl)pentanamide | N-[2-(diethylamino)ethyl]-5-(8-methoxy-4-oxo-2-sulfanylidene-1,5-dihydropyrimido[5,4-b]indol-3-yl)pentanamide | N-[2-(diethylamino)ethyl]-5-(8-methoxy-4-oxo-2-thioxo-1,2,4,5-tetrahydro-3H-pyrimido[5,4-b]indol-3-yl)pentanamide | SMR000028552 | cid_3244883 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H31N5O3S |
|---|
| Mol. Mass. | 445.578 |
|---|
| SMILES | CCN(CC)CCNC(=O)CCCCn1c(=S)[nH]c2c3cc(OC)ccc3[nH]c2c1=O |
|---|
| Structure | 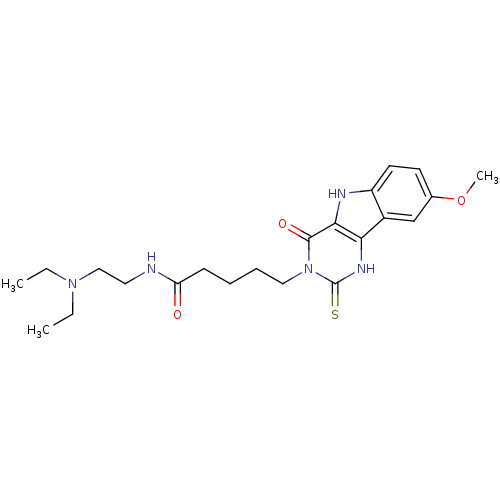
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 PubChem, PC Dose Response confirmation of small molecule APOBEC3G DNA Deaminase Inhibitors via a fluorescence-based single-stranded DNA deaminase assay PubChem Bioassay(2011)[AID]
PubChem, PC Dose Response confirmation of small molecule APOBEC3G DNA Deaminase Inhibitors via a fluorescence-based single-stranded DNA deaminase assay PubChem Bioassay(2011)[AID]