| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Serine/threonine-protein kinase TBK1 |
|---|
| Ligand | BDBM357857 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1803718 (CHEMBL4276010) |
|---|
| IC50 | 851±n/a nM |
|---|
| Citation |  Beyett, TS; Gan, X; Reilly, SM; Gomez, AV; Chang, L; Tesmer, JJG; Saltiel, AR; Showalter, HD Design, synthesis, and biological activity of substituted 2-amino-5-oxo-5H-chromeno[2,3-b]pyridine-3-carboxylic acid derivatives as inhibitors of the inflammatory kinases TBK1 and IKK? for the treatment of obesity. Bioorg Med Chem26:5443-5461 (2018) [PubMed] Article Beyett, TS; Gan, X; Reilly, SM; Gomez, AV; Chang, L; Tesmer, JJG; Saltiel, AR; Showalter, HD Design, synthesis, and biological activity of substituted 2-amino-5-oxo-5H-chromeno[2,3-b]pyridine-3-carboxylic acid derivatives as inhibitors of the inflammatory kinases TBK1 and IKK? for the treatment of obesity. Bioorg Med Chem26:5443-5461 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Serine/threonine-protein kinase TBK1 |
|---|
| Name: | Serine/threonine-protein kinase TBK1 |
|---|
| Synonyms: | NAK | NF-kappa-B-activating kinase | Protein cereblon/Serine/threonine-protein kinase TBK1 | T2K | TANK-binding kinase 1 (TBK-1) | TANK-binding kinase 1 (TBK1) | TBK1 | TBK1_HUMAN |
|---|
| Type: | protein |
|---|
| Mol. Mass.: | 83645.20 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q9UHD2 |
|---|
| Residue: | 729 |
|---|
| Sequence: | MQSTSNHLWLLSDILGQGATANVFRGRHKKTGDLFAIKVFNNISFLRPVDVQMREFEVLK
KLNHKNIVKLFAIEEETTTRHKVLIMEFCPCGSLYTVLEEPSNAYGLPESEFLIVLRDVV
GGMNHLRENGIVHRDIKPGNIMRVIGEDGQSVYKLTDFGAARELEDDEQFVSLYGTEEYL
HPDMYERAVLRKDHQKKYGATVDLWSIGVTFYHAATGSLPFRPFEGPRRNKEVMYKIITG
KPSGAISGVQKAENGPIDWSGDMPVSCSLSRGLQVLLTPVLANILEADQEKCWGFDQFFA
ETSDILHRMVIHVFSLQQMTAHKIYIHSYNTATIFHELVYKQTKIISSNQELIYEGRRLV
LEPGRLAQHFPKTTEENPIFVVSREPLNTIGLIYEKISLPKVHPRYDLDGDASMAKAITG
VVCYACRIASTLLLYQELMRKGIRWLIELIKDDYNETVHKKTEVVITLDFCIRNIEKTVK
VYEKLMKINLEAAELGEISDIHTKLLRLSSSQGTIETSLQDIDSRLSPGGSLADAWAHQE
GTHPKDRNVEKLQVLLNCMTEIYYQFKKDKAERRLAYNEEQIHKFDKQKLYYHATKAMTH
FTDECVKKYEAFLNKSEEWIRKMLHLRKQLLSLTNQCFDIEEEVSKYQEYTNELQETLPQ
KMFTASSGIKHTMTPIYPSSNTLVEMTLGMKKLKEEMEGVVKELAENNHILERFGSLTMD
GGLRNVDCL
|
|
|
|---|
| BDBM357857 |
|---|
| n/a |
|---|
| Name | BDBM357857 |
|---|
| Synonyms: | Aphthasol | US10214536, Amlexanox |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H14N2O4 |
|---|
| Mol. Mass. | 298.2934 |
|---|
| SMILES | CC(C)c1ccc2oc3nc(N)c(cc3c(=O)c2c1)C(O)=O |
|---|
| Structure | 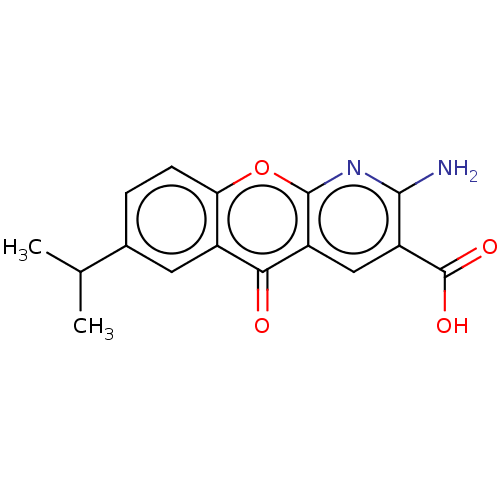
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Beyett, TS; Gan, X; Reilly, SM; Gomez, AV; Chang, L; Tesmer, JJG; Saltiel, AR; Showalter, HD Design, synthesis, and biological activity of substituted 2-amino-5-oxo-5H-chromeno[2,3-b]pyridine-3-carboxylic acid derivatives as inhibitors of the inflammatory kinases TBK1 and IKK? for the treatment of obesity. Bioorg Med Chem26:5443-5461 (2018) [PubMed] Article
Beyett, TS; Gan, X; Reilly, SM; Gomez, AV; Chang, L; Tesmer, JJG; Saltiel, AR; Showalter, HD Design, synthesis, and biological activity of substituted 2-amino-5-oxo-5H-chromeno[2,3-b]pyridine-3-carboxylic acid derivatives as inhibitors of the inflammatory kinases TBK1 and IKK? for the treatment of obesity. Bioorg Med Chem26:5443-5461 (2018) [PubMed] Article