| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM223287 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1876813 (CHEMBL4378207) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Munck Af Rosensch�ld, M; Johannesson, P; Nikitidis, A; Tyrchan, C; Chang, HF; R�nn, R; Chapman, D; Ullah, V; Nikitidis, G; Glader, P; K�ck, H; Bonn, B; W�gberg, F; Bj�rkstrand, E; Andersson, U; Swedin, L; Rohman, M; Andreasson, T; Bergstr�m, EL; Jiang, F; Zhou, XH; Lundqvist, AJ; Malmberg, A; Ek, M; Gordon, E; Pettersen, A; Ripa, L; Davis, AM Discovery of the Oral Leukotriene C4 Synthase Inhibitor (1 J Med Chem62:7769-7787 (2019) [PubMed] Article Munck Af Rosensch�ld, M; Johannesson, P; Nikitidis, A; Tyrchan, C; Chang, HF; R�nn, R; Chapman, D; Ullah, V; Nikitidis, G; Glader, P; K�ck, H; Bonn, B; W�gberg, F; Bj�rkstrand, E; Andersson, U; Swedin, L; Rohman, M; Andreasson, T; Bergstr�m, EL; Jiang, F; Zhou, XH; Lundqvist, AJ; Malmberg, A; Ek, M; Gordon, E; Pettersen, A; Ripa, L; Davis, AM Discovery of the Oral Leukotriene C4 Synthase Inhibitor (1 J Med Chem62:7769-7787 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM223287 |
|---|
| n/a |
|---|
| Name | BDBM223287 |
|---|
| Synonyms: | (1S,2S)-2-({5-[(5-Chloro-2,4-difluorophenyl)(2-fluoro-2-methylpropyl)amino]-3-methoxypyrazin-2-yl}carbonyl)cyclopropanecarboxylic acid | US20160326143, 62 | US9657001, 62 |
|---|
| Type | Small Organic Molecule |
|---|
| Emp. Form. | C20H19ClF3N3O4 |
|---|
| Mol. Mass. | 457.831 |
|---|
| SMILES | COc1nc(cnc1C(=O)[C@H]1C[C@@H]1C(O)=O)N(CC(C)(C)F)c1cc(Cl)c(F)cc1F |
|---|
| Structure | 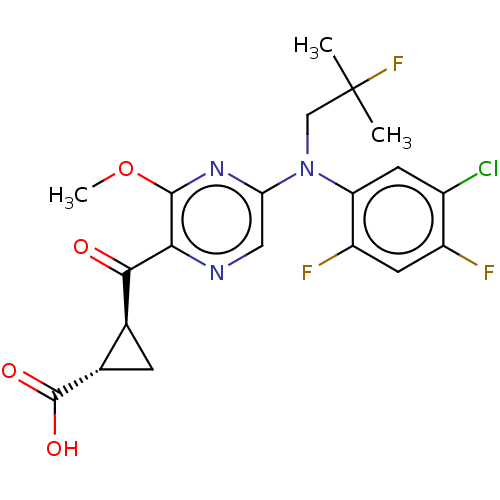
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Munck Af Rosensch�ld, M; Johannesson, P; Nikitidis, A; Tyrchan, C; Chang, HF; R�nn, R; Chapman, D; Ullah, V; Nikitidis, G; Glader, P; K�ck, H; Bonn, B; W�gberg, F; Bj�rkstrand, E; Andersson, U; Swedin, L; Rohman, M; Andreasson, T; Bergstr�m, EL; Jiang, F; Zhou, XH; Lundqvist, AJ; Malmberg, A; Ek, M; Gordon, E; Pettersen, A; Ripa, L; Davis, AM Discovery of the Oral Leukotriene C4 Synthase Inhibitor (1 J Med Chem62:7769-7787 (2019) [PubMed] Article
Munck Af Rosensch�ld, M; Johannesson, P; Nikitidis, A; Tyrchan, C; Chang, HF; R�nn, R; Chapman, D; Ullah, V; Nikitidis, G; Glader, P; K�ck, H; Bonn, B; W�gberg, F; Bj�rkstrand, E; Andersson, U; Swedin, L; Rohman, M; Andreasson, T; Bergstr�m, EL; Jiang, F; Zhou, XH; Lundqvist, AJ; Malmberg, A; Ek, M; Gordon, E; Pettersen, A; Ripa, L; Davis, AM Discovery of the Oral Leukotriene C4 Synthase Inhibitor (1 J Med Chem62:7769-7787 (2019) [PubMed] Article