| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C19 |
|---|
| Ligand | BDBM50519571 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1876811 (CHEMBL4378205) |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Munck Af Rosensch�ld, M; Johannesson, P; Nikitidis, A; Tyrchan, C; Chang, HF; R�nn, R; Chapman, D; Ullah, V; Nikitidis, G; Glader, P; K�ck, H; Bonn, B; W�gberg, F; Bj�rkstrand, E; Andersson, U; Swedin, L; Rohman, M; Andreasson, T; Bergstr�m, EL; Jiang, F; Zhou, XH; Lundqvist, AJ; Malmberg, A; Ek, M; Gordon, E; Pettersen, A; Ripa, L; Davis, AM Discovery of the Oral Leukotriene C4 Synthase Inhibitor (1 J Med Chem62:7769-7787 (2019) [PubMed] Article Munck Af Rosensch�ld, M; Johannesson, P; Nikitidis, A; Tyrchan, C; Chang, HF; R�nn, R; Chapman, D; Ullah, V; Nikitidis, G; Glader, P; K�ck, H; Bonn, B; W�gberg, F; Bj�rkstrand, E; Andersson, U; Swedin, L; Rohman, M; Andreasson, T; Bergstr�m, EL; Jiang, F; Zhou, XH; Lundqvist, AJ; Malmberg, A; Ek, M; Gordon, E; Pettersen, A; Ripa, L; Davis, AM Discovery of the Oral Leukotriene C4 Synthase Inhibitor (1 J Med Chem62:7769-7787 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C19 |
|---|
| Name: | Cytochrome P450 2C19 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | (S)-limonene 7-monooxygenase | CP2CJ_HUMAN | CYP2C19 | CYPIIC17 | CYPIIC19 | Cytochrome P450 2C19 (CYP2C19) | Cytochrome P450 2C19 [I331V] | Cytochrome P450-11A | Cytochrome P450-254C | Fenbendazole monooxygenase (4'-hydroxylating) | Mephenytoin 4-hydroxylase | P450-11A | P450-254C |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55935.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P33261 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDPFVVLVLCLSCLLLLSIWRQSSGRGKLPPGPTPLPVIGNILQIDIKDVSKSLTNLSKI
YGPVFTLYFGLERMVVLHGYEVVKEALIDLGEEFSGRGHFPLAERANRGFGIVFSNGKRW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFQKRFDYKDQQFLNLMEKLNENIRIVSTPWIQICNNFPTIIDYFPGTHNKLLKNLAFM
ESDILEKVKEHQESMDINNPRDFIDCFLIKMEKEKQNQQSEFTIENLVITAADLLGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRGHMPYTDAVVHEVQRYID
LIPTSLPHAVTCDVKFRNYLIPKGTTILTSLTSVLHDNKEFPNPEMFDPRHFLDEGGNFK
KSNYFMPFSAGKRICVGEGLARMELFLFLTFILQNFNLKSLIDPKDLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50519571 |
|---|
| n/a |
|---|
| Name | BDBM50519571 |
|---|
| Synonyms: | CHEMBL4454609 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H23ClN2O4 |
|---|
| Mol. Mass. | 450.914 |
|---|
| SMILES | COc1cc(cnc1C(=O)[C@H]1C[C@@H]1C(O)=O)N(CC1CC1)c1ccc(Cl)c2ccccc12 |r| |
|---|
| Structure | 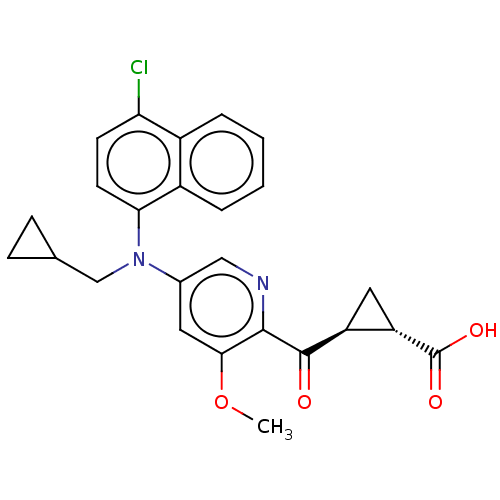
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Munck Af Rosensch�ld, M; Johannesson, P; Nikitidis, A; Tyrchan, C; Chang, HF; R�nn, R; Chapman, D; Ullah, V; Nikitidis, G; Glader, P; K�ck, H; Bonn, B; W�gberg, F; Bj�rkstrand, E; Andersson, U; Swedin, L; Rohman, M; Andreasson, T; Bergstr�m, EL; Jiang, F; Zhou, XH; Lundqvist, AJ; Malmberg, A; Ek, M; Gordon, E; Pettersen, A; Ripa, L; Davis, AM Discovery of the Oral Leukotriene C4 Synthase Inhibitor (1 J Med Chem62:7769-7787 (2019) [PubMed] Article
Munck Af Rosensch�ld, M; Johannesson, P; Nikitidis, A; Tyrchan, C; Chang, HF; R�nn, R; Chapman, D; Ullah, V; Nikitidis, G; Glader, P; K�ck, H; Bonn, B; W�gberg, F; Bj�rkstrand, E; Andersson, U; Swedin, L; Rohman, M; Andreasson, T; Bergstr�m, EL; Jiang, F; Zhou, XH; Lundqvist, AJ; Malmberg, A; Ek, M; Gordon, E; Pettersen, A; Ripa, L; Davis, AM Discovery of the Oral Leukotriene C4 Synthase Inhibitor (1 J Med Chem62:7769-7787 (2019) [PubMed] Article