| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM85097 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_51355 (CHEMBL666156) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Bromidge, SM; Dabbs, S; Davies, DT; Davies, S; Duckworth, DM; Forbes, IT; Gaster, LM; Ham, P; Jones, GE; King, FD; Mulholland, KR; Saunders, DV; Wyman, PA; Blaney, FE; Clarke, SE; Blackburn, TP; Holland, V; Kennett, GA; Lightowler, S; Middlemiss, DN; Trail, B; Riley, GJ; Wood, MD Biarylcarbamoylindolines are novel and selective 5-HT(2C) receptor inverse agonists: identification of 5-methyl-1-[[2-[(2-methyl-3-pyridyl)oxy]- 5-pyridyl]carbamoyl]-6-trifluoromethylindoline (SB-243213) as a potential antidepressant/anxiolytic agent. J Med Chem43:1123-34 (2000) [PubMed] Bromidge, SM; Dabbs, S; Davies, DT; Davies, S; Duckworth, DM; Forbes, IT; Gaster, LM; Ham, P; Jones, GE; King, FD; Mulholland, KR; Saunders, DV; Wyman, PA; Blaney, FE; Clarke, SE; Blackburn, TP; Holland, V; Kennett, GA; Lightowler, S; Middlemiss, DN; Trail, B; Riley, GJ; Wood, MD Biarylcarbamoylindolines are novel and selective 5-HT(2C) receptor inverse agonists: identification of 5-methyl-1-[[2-[(2-methyl-3-pyridyl)oxy]- 5-pyridyl]carbamoyl]-6-trifluoromethylindoline (SB-243213) as a potential antidepressant/anxiolytic agent. J Med Chem43:1123-34 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM85097 |
|---|
| n/a |
|---|
| Name | BDBM85097 |
|---|
| Synonyms: | CAS_181632-25-7 | CHEMBL14563 | SB 242084 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H19ClN4O2 |
|---|
| Mol. Mass. | 394.854 |
|---|
| SMILES | Cc1cc2CCN(C(=O)Nc3ccc(Oc4cccnc4C)nc3)c2cc1Cl |
|---|
| Structure | 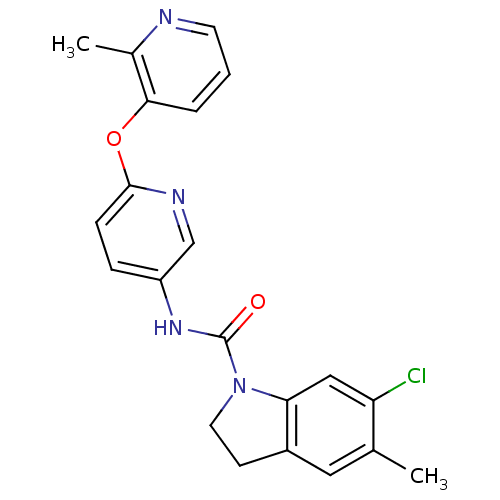
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bromidge, SM; Dabbs, S; Davies, DT; Davies, S; Duckworth, DM; Forbes, IT; Gaster, LM; Ham, P; Jones, GE; King, FD; Mulholland, KR; Saunders, DV; Wyman, PA; Blaney, FE; Clarke, SE; Blackburn, TP; Holland, V; Kennett, GA; Lightowler, S; Middlemiss, DN; Trail, B; Riley, GJ; Wood, MD Biarylcarbamoylindolines are novel and selective 5-HT(2C) receptor inverse agonists: identification of 5-methyl-1-[[2-[(2-methyl-3-pyridyl)oxy]- 5-pyridyl]carbamoyl]-6-trifluoromethylindoline (SB-243213) as a potential antidepressant/anxiolytic agent. J Med Chem43:1123-34 (2000) [PubMed]
Bromidge, SM; Dabbs, S; Davies, DT; Davies, S; Duckworth, DM; Forbes, IT; Gaster, LM; Ham, P; Jones, GE; King, FD; Mulholland, KR; Saunders, DV; Wyman, PA; Blaney, FE; Clarke, SE; Blackburn, TP; Holland, V; Kennett, GA; Lightowler, S; Middlemiss, DN; Trail, B; Riley, GJ; Wood, MD Biarylcarbamoylindolines are novel and selective 5-HT(2C) receptor inverse agonists: identification of 5-methyl-1-[[2-[(2-methyl-3-pyridyl)oxy]- 5-pyridyl]carbamoyl]-6-trifluoromethylindoline (SB-243213) as a potential antidepressant/anxiolytic agent. J Med Chem43:1123-34 (2000) [PubMed]