

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50143314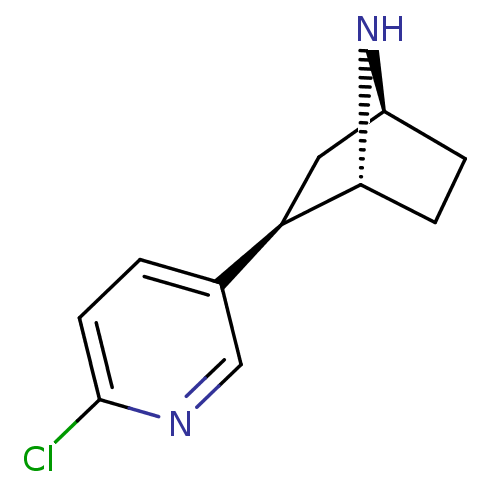 ((+)-Epibatidine | (-)-epibatidine | (1R,2R,4S)-2-(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]epibatidine binding at the nicotinic acetylcholine receptor alpha4-beta2 in male rat cerebral cortex | J Med Chem 44: 2229-37 (2001) BindingDB Entry DOI: 10.7270/Q2BV7HVD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50100717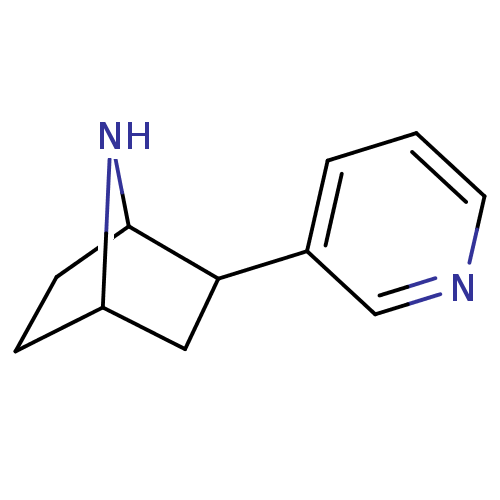 (2-(pyridin-3-yl)-7-aza-bicyclo[2.2.1]heptane | 2-P...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]epibatidine binding at the nicotinic acetylcholine receptor alpha4-beta2 in male rat cerebral cortex | J Med Chem 44: 2229-37 (2001) BindingDB Entry DOI: 10.7270/Q2BV7HVD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50100715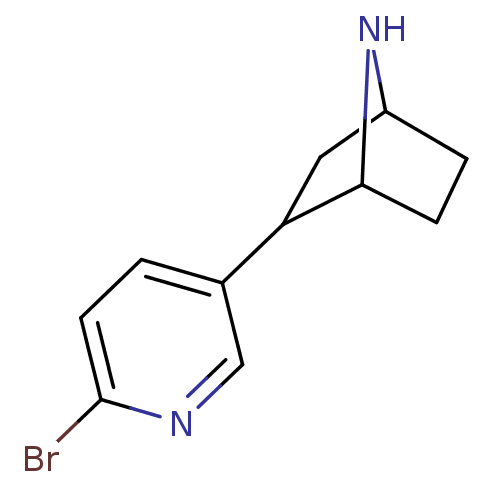 (2-(6-Bromo-pyridin-3-yl)-7-aza-bicyclo[2.2.1]hepta...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]epibatidine binding at the nicotinic acetylcholine receptor alpha4-beta2 in male rat cerebral cortex | J Med Chem 44: 2229-37 (2001) BindingDB Entry DOI: 10.7270/Q2BV7HVD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50143320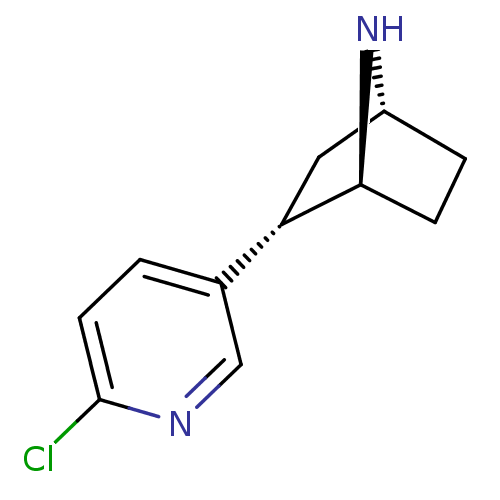 ((+)-epibatidine | (-)-1-epidatidine | (1S,2S,4R)-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]epibatidine binding at the nicotinic acetylcholine receptor alpha4-beta2 in male rat cerebral cortex | J Med Chem 44: 2229-37 (2001) BindingDB Entry DOI: 10.7270/Q2BV7HVD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50100707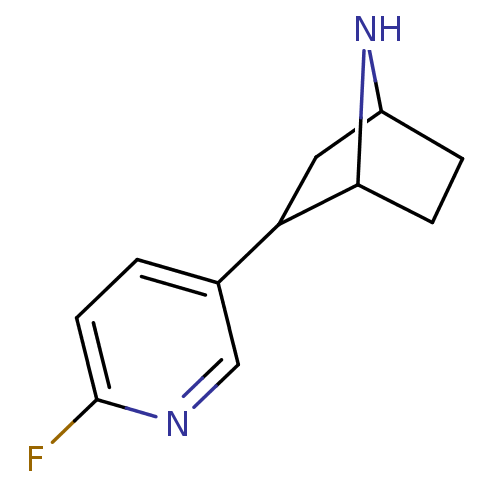 ((R)-2-(6-Fluoro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.0270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]epibatidine binding at the nicotinic acetylcholine receptor alpha4-beta2 in male rat cerebral cortex | J Med Chem 44: 2229-37 (2001) BindingDB Entry DOI: 10.7270/Q2BV7HVD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50011851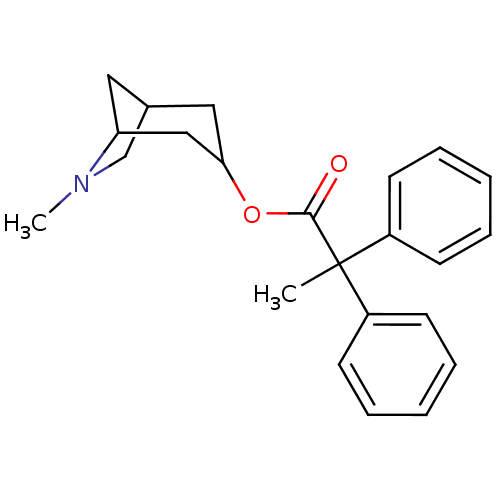 (2,2-Diphenyl-propionic acid 6-methyl-6-aza-bicyclo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding against muscarinic acetylcholine receptor in rat brain. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50100717 (2-(pyridin-3-yl)-7-aza-bicyclo[2.2.1]heptane | 2-P...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.0310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Non-specific binding in presence of 300 uM nicotine at nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex membranes | J Med Chem 44: 2229-37 (2001) BindingDB Entry DOI: 10.7270/Q2BV7HVD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (RAT) | BDBM50011851 (2,2-Diphenyl-propionic acid 6-methyl-6-aza-bicyclo...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0383 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description The compound was tested for binding activity against muscarinic acetylcholine receptor M1, using [3H]QNB as the radioligand. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50143320 ((+)-epibatidine | (-)-1-epidatidine | (1S,2S,4R)-2...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Non-specific binding in presence of 300 uM nicotine at nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex membranes | J Med Chem 44: 2229-37 (2001) BindingDB Entry DOI: 10.7270/Q2BV7HVD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (Homo sapiens (Human)) | BDBM50011851 (2,2-Diphenyl-propionic acid 6-methyl-6-aza-bicyclo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0540 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding against muscarinic acetylcholine receptor in guinea pig ileum | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50143314 ((+)-Epibatidine | (-)-epibatidine | (1R,2R,4S)-2-(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.0580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Non-specific binding in presence of 300 uM nicotine at nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex membranes | J Med Chem 44: 2229-37 (2001) BindingDB Entry DOI: 10.7270/Q2BV7HVD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50100712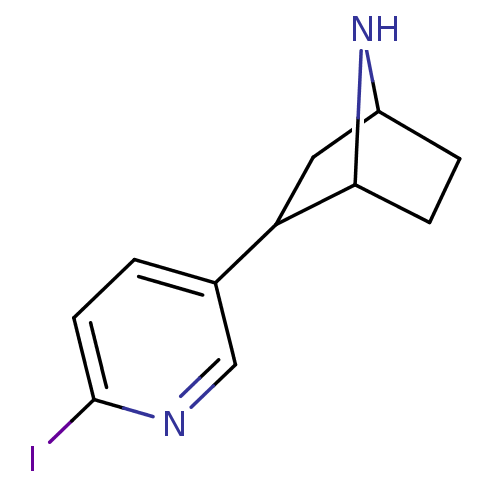 (2-(6-Iodo-pyridin-3-yl)-7-aza-bicyclo[2.2.1]heptan...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]epibatidine binding at the nicotinic acetylcholine receptor alpha4-beta2 in male rat cerebral cortex | J Med Chem 44: 2229-37 (2001) BindingDB Entry DOI: 10.7270/Q2BV7HVD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50011851 (2,2-Diphenyl-propionic acid 6-methyl-6-aza-bicyclo...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0880 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding to CHO cells bearing transfected muscarinic acetylcholine receptor M1 | J Med Chem 34: 1436-40 (1991) BindingDB Entry DOI: 10.7270/Q2QC043X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (RAT) | BDBM50011851 (2,2-Diphenyl-propionic acid 6-methyl-6-aza-bicyclo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0986 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description The compound was tested for binding activity against muscarinic acetylcholine receptor M3, using [3H]-QNB as the radioligand. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 1A (Rattus norvegicus (rat)) | BDBM82368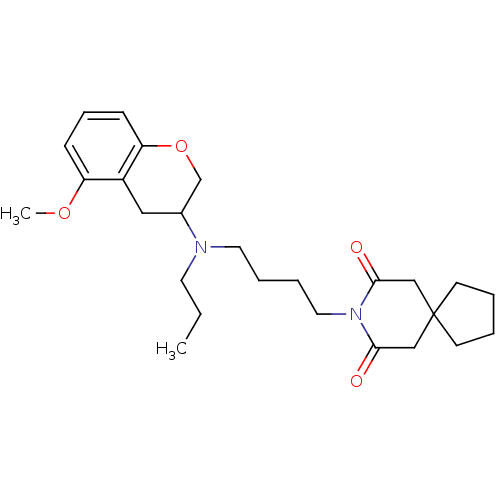 (CAS_121851 | CHEMBL11592 | CHEMBL301060 | NSC_1218...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lausanne Curated by ChEMBL | Assay Description Binding affinity of a compound to rat brain 5-hydroxytryptamine 1A (serotonin) receptor assayed by radiolabeled [3H]-8-OH-DPAT ligand displacement | J Med Chem 39: 126-34 (1996) Article DOI: 10.1021/jm950410b BindingDB Entry DOI: 10.7270/Q2VX0K7V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone-lysine N-methyltransferase EZH2 (Homo sapiens (Human)) | BDBM50246967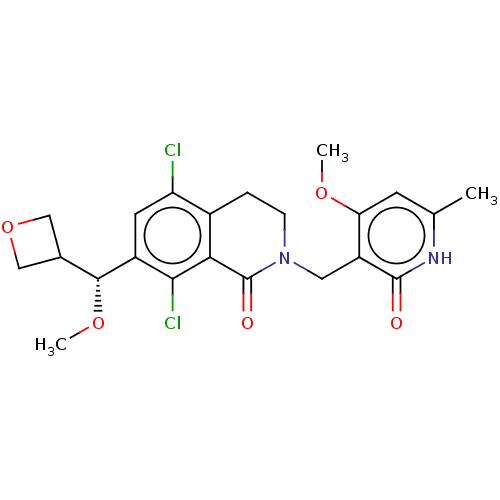 (CHEMBL4080228 | US10570121, Example 81) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | MCE PC cid PC sid PDB UniChem | PDB Article PubMed | <0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
WuXi AppTec Curated by ChEMBL | Assay Description Binding affinity to EZH2 (unknown origin) | J Med Chem 61: 650-665 (2018) Article DOI: 10.1021/acs.jmedchem.7b01375 BindingDB Entry DOI: 10.7270/Q2X069G8 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histone-lysine N-methyltransferase EZH2 (Homo sapiens (Human)) | BDBM50246967 (CHEMBL4080228 | US10570121, Example 81) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | MCE PC cid PC sid PDB UniChem | PDB Article PubMed | <0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
WuXi AppTec Curated by ChEMBL | Assay Description Binding affinity to EZH2 (unknown origin) | J Med Chem 61: 650-665 (2018) Article DOI: 10.1021/acs.jmedchem.7b01375 BindingDB Entry DOI: 10.7270/Q2X069G8 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50011851 (2,2-Diphenyl-propionic acid 6-methyl-6-aza-bicyclo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.118 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]pirenzepine binding against muscarinic acetylcholine receptor in rat brain. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50104923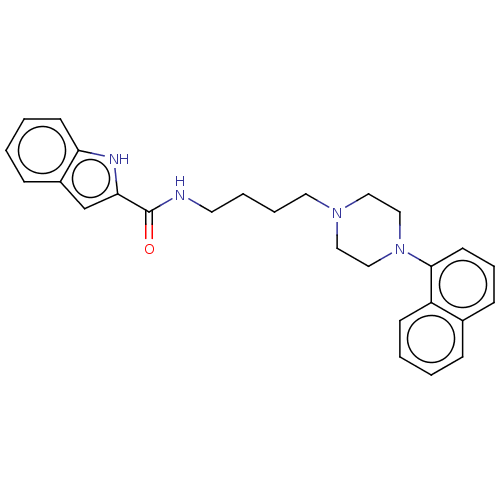 (CHEMBL3597643) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.118 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute on Drug Abuse- Intramural Research Program Curated by ChEMBL | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D3 receptor (unknown origin) expressed in HEK293 cells after 1 hr by liquid scintillation ... | J Med Chem 58: 6195-213 (2015) Article DOI: 10.1021/acs.jmedchem.5b00776 BindingDB Entry DOI: 10.7270/Q2KS6T92 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50105009 (CHEMBL3597645) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.128 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute on Drug Abuse- Intramural Research Program Curated by ChEMBL | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D3 receptor (unknown origin) expressed in HEK293 cells after 1 hr by liquid scintillation ... | J Med Chem 58: 6195-213 (2015) Article DOI: 10.1021/acs.jmedchem.5b00776 BindingDB Entry DOI: 10.7270/Q2KS6T92 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50368152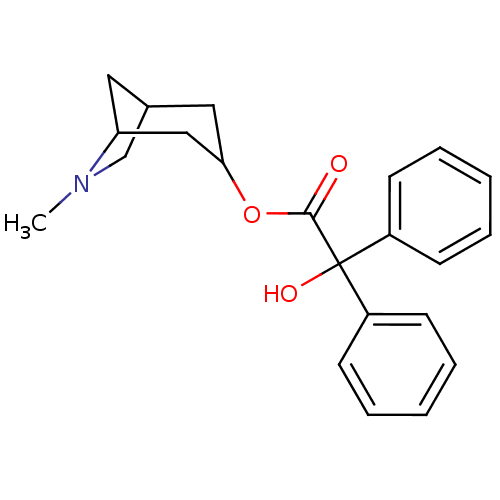 (CHEMBL318812) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.166 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]pirenzepine binding against muscarinic acetylcholine receptor in rat brain. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50266969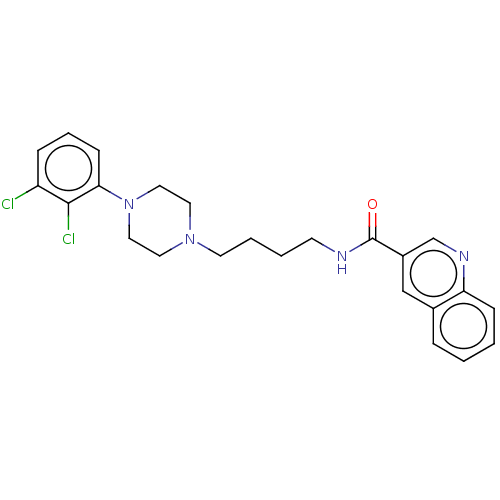 (CHEMBL4093656) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 0.172 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute on Drug Abuse-Intramural Research Program Curated by ChEMBL | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D3 receptor expressed in HEK293 cells after 1 hr by MicroBeta microplate counting method | J Med Chem 60: 580-593 (2017) Article DOI: 10.1021/acs.jmedchem.6b01148 BindingDB Entry DOI: 10.7270/Q2PV6NVS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50104998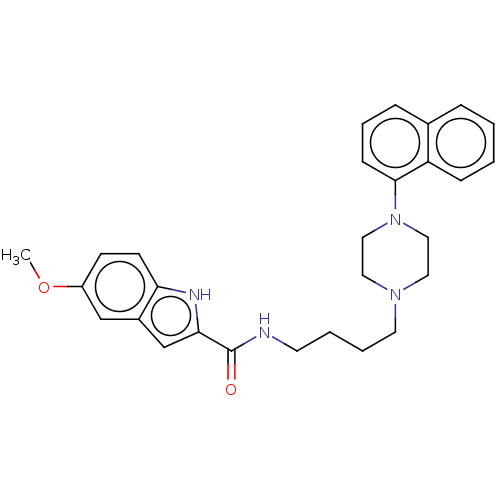 (CHEMBL3597644) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.173 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute on Drug Abuse- Intramural Research Program Curated by ChEMBL | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D3 receptor (unknown origin) expressed in HEK293 cells after 1 hr by liquid scintillation ... | J Med Chem 58: 6195-213 (2015) Article DOI: 10.1021/acs.jmedchem.5b00776 BindingDB Entry DOI: 10.7270/Q2KS6T92 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M2 (RAT) | BDBM50368152 (CHEMBL318812) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.177 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Compound was tested for binding activity against rat muscarinic acetylcholine receptor M2 using [3H]QNB as the radioligand | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol phosphorylceramide synthase (Candida albicans) | BDBM50408926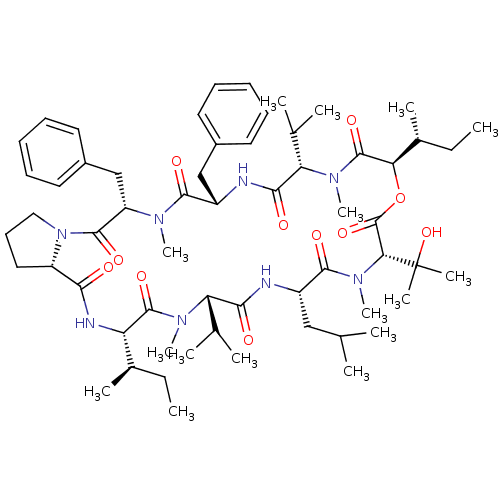 (Aureobasidin A | CHEMBL1793802) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacia Corp. Curated by ChEMBL | Assay Description Inhibition of Candida albicans ATCC 38247 inositol phosphorylceramide synthase preincubated for 30 mins | Antimicrob Agents Chemother 53: 496-504 (2009) Article DOI: 10.1128/AAC.00633-08 BindingDB Entry DOI: 10.7270/Q27H1JV5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50001023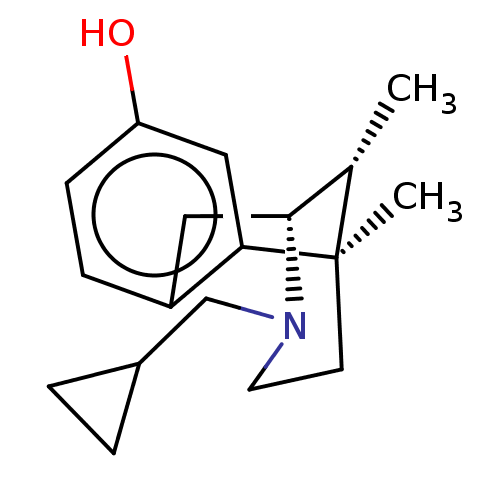 ((2R,6R,11R)-3-Cyclopropylmethyl-6,11-dimethyl-1,2,...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Binding affinity against opioid receptor mu using [3H]-DAMGO as radioligand. | J Med Chem 35: 2812-8 (1992) BindingDB Entry DOI: 10.7270/Q29W0G3H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50011851 (2,2-Diphenyl-propionic acid 6-methyl-6-aza-bicyclo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.196 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding against muscarinic acetylcholine receptor in rat heart. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50368152 (CHEMBL318812) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.197 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]pirenzepine binding against muscarinic acetylcholine receptor in rat brain. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 1A (Rattus norvegicus (rat)) | BDBM50036874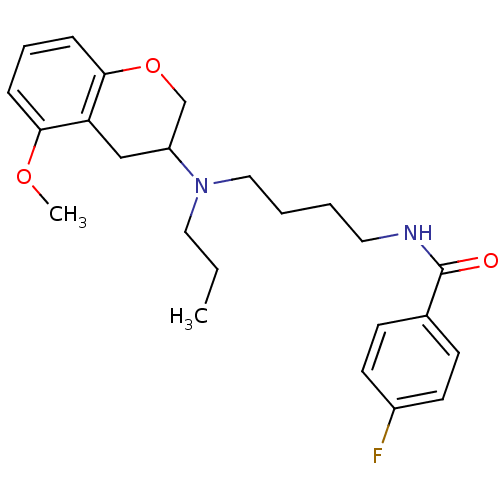 (4-Fluoro-N-{4-[(5-methoxy-chroman-3-yl)-propyl-ami...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lausanne Curated by ChEMBL | Assay Description Binding affinity of a compound to rat brain 5-hydroxytryptamine 1A (serotonin) receptor assayed by radiolabeled [3H]-8-OH-DPAT ligand displacement | J Med Chem 39: 126-34 (1996) Article DOI: 10.1021/jm950410b BindingDB Entry DOI: 10.7270/Q2VX0K7V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Rattus norvegicus (rat)) | BDBM50001019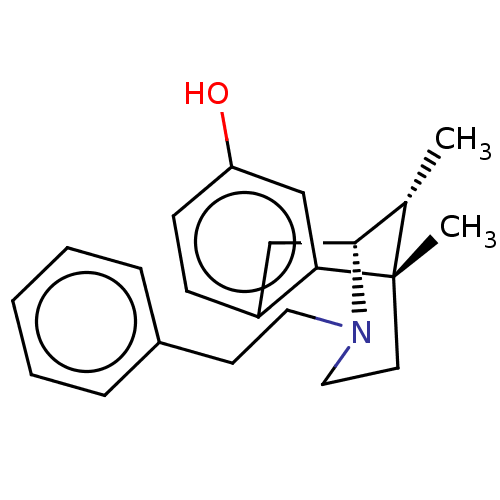 (6,11-Dimethyl-3-phenethyl-1,2,3,4,5,6-hexahydro-2,...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Binding affinity against opioid receptor mu using [3H]-DAMGO as radioligand. | J Med Chem 35: 2812-8 (1992) BindingDB Entry DOI: 10.7270/Q29W0G3H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Homo sapiens (Human)) | BDBM50397944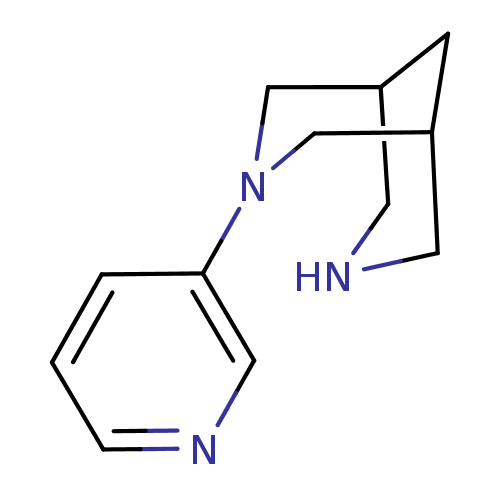 (CHEMBL2177512) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Targacept, Inc. Curated by ChEMBL | Assay Description Binding affinity to alpha4beta2 nAChR | J Med Chem 55: 9181-94 (2012) Article DOI: 10.1021/jm3006542 BindingDB Entry DOI: 10.7270/Q2W0972W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (RAT) | BDBM50368152 (CHEMBL318812) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Binding activity against rat muscarinic acetylcholine receptor M1 using [3H]QNB as the radioligand | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50368152 (CHEMBL318812) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.224 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding against muscarinic acetylcholine receptor in rat brain. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Inositol phosphorylceramide synthase catalytic subunit AUR1 (Saccharomyces cerevisiae S288c) | BDBM50408926 (Aureobasidin A | CHEMBL1793802) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacia Corp. Curated by ChEMBL | Assay Description Inhibition of Saccharomyces cerevisiae SJ21R inositol phosphorylceramide synthase preincubated for 30 mins | Antimicrob Agents Chemother 53: 496-504 (2009) Article DOI: 10.1128/AAC.00633-08 BindingDB Entry DOI: 10.7270/Q27H1JV5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sodium-dependent serotonin transporter (Rattus norvegicus (rat)) | BDBM50051753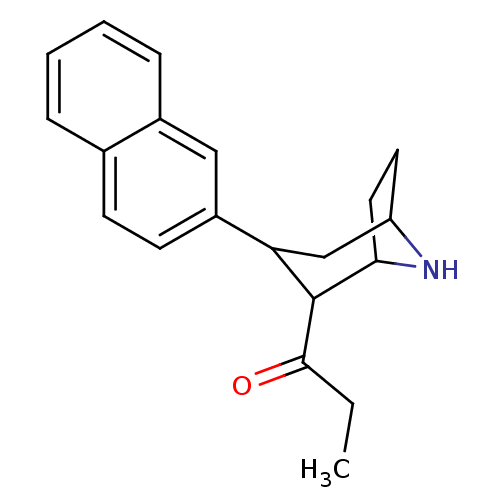 (1-(3-Naphthalen-2-yl-8-aza-bicyclo[3.2.1]oct-2-yl)...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York Curated by ChEMBL | Assay Description Affinity for the displacement of [3H]-paroxetine binding to serotonin transporter (SERT) in rat frontal cortex membranes | J Med Chem 44: 1509-15 (2001) BindingDB Entry DOI: 10.7270/Q2H1318R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50105004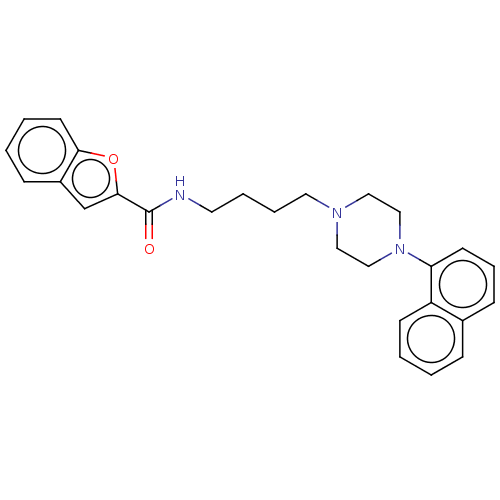 (CHEMBL3596212) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.244 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute on Drug Abuse- Intramural Research Program Curated by ChEMBL | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D3 receptor (unknown origin) expressed in HEK293 cells after 1 hr by liquid scintillation ... | J Med Chem 58: 6195-213 (2015) Article DOI: 10.1021/acs.jmedchem.5b00776 BindingDB Entry DOI: 10.7270/Q2KS6T92 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 1A (Rattus norvegicus (rat)) | BDBM50039822 (1-(2-Cyclohexyl-ethyl)-4-(2,3-dihydro-benzo[1,4]di...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lausanne Curated by ChEMBL | Assay Description Binding affinity of a compound to rat brain 5-hydroxytryptamine 1A (serotonin) receptor assayed by radiolabeled [3H]-8-OH-DPAT ligand displacement | J Med Chem 39: 126-34 (1996) Article DOI: 10.1021/jm950410b BindingDB Entry DOI: 10.7270/Q2VX0K7V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50368152 (CHEMBL318812) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.254 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding against muscarinic acetylcholine receptor in rat brain. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (Homo sapiens (Human)) | BDBM50011851 (2,2-Diphenyl-propionic acid 6-methyl-6-aza-bicyclo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibit the binding of [N-mnethyl-3H]-scopolamine [3H]-NMS) to Muscarinic acetylcholine receptor of human IRM-30 neuroblastoma cells | J Med Chem 30: 805-9 (1987) BindingDB Entry DOI: 10.7270/Q2B27XHZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Homo sapiens (Human)) | BDBM50135597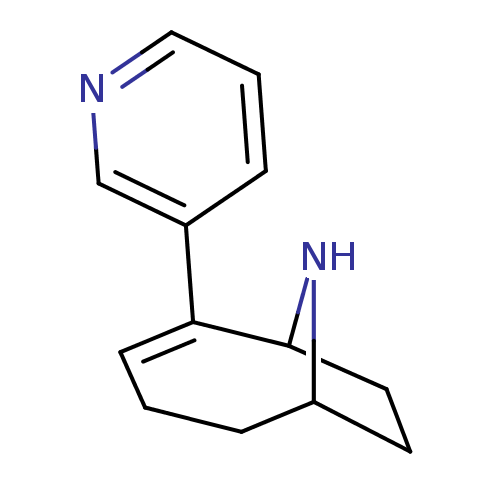 (2-Pyridin-3-yl-9-aza-bicyclo[4.2.1]non-2-ene | CHE...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Targacept, Inc. Curated by ChEMBL | Assay Description Binding affinity to alpha4beta2 nAChR | J Med Chem 55: 9181-94 (2012) Article DOI: 10.1021/jm3006542 BindingDB Entry DOI: 10.7270/Q2W0972W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neprilysin (Homo sapiens (Human)) | BDBM50411736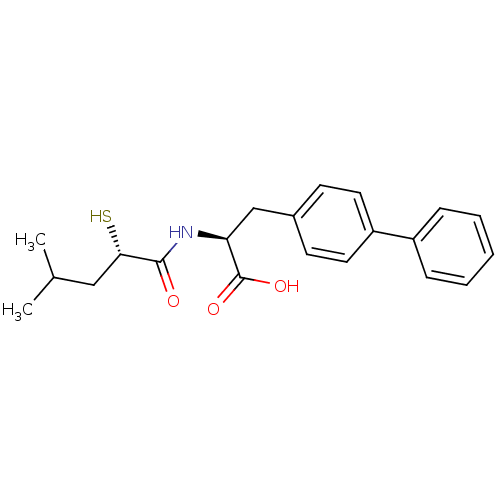 (CHEMBL271225) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of human recombinant NEP by fluorescence assay | Bioorg Med Chem Lett 18: 732-7 (2008) Article DOI: 10.1016/j.bmcl.2007.11.048 BindingDB Entry DOI: 10.7270/Q2GT5PCB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50129425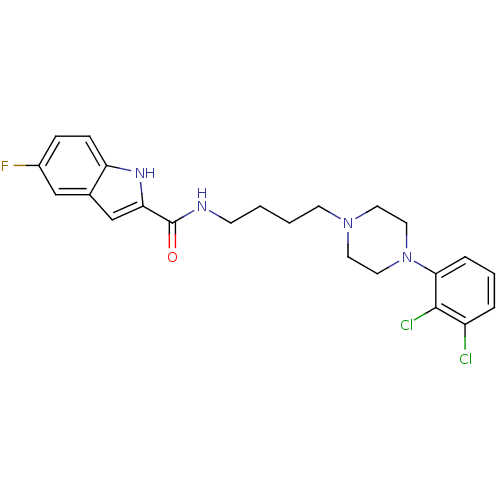 (5-Fluoro-1H-indole-2-carboxylic acid {4-[4-(2,3-di...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute on Drug Abuse- Intramural Research Program Curated by ChEMBL | Assay Description Displacement of [3H]-N-methylspiperone from human dopamine D3 receptor (unknown origin) expressed in HEK293 cells after 1 hr by liquid scintillation ... | J Med Chem 58: 6195-213 (2015) Article DOI: 10.1021/acs.jmedchem.5b00776 BindingDB Entry DOI: 10.7270/Q2KS6T92 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 1A (Rattus norvegicus (rat)) | BDBM21392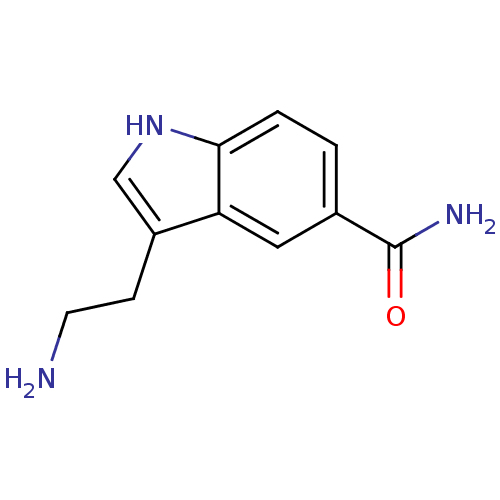 (3-(2-aminoethyl)-1H-indole-5-carboxamide | 5-CT | ...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.295 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Lausanne Curated by ChEMBL | Assay Description Binding affinity of a compound to rat brain 5-hydroxytryptamine 1A (serotonin) receptor assayed by radiolabeled [3H]-8-OH-DPAT ligand displacement | J Med Chem 39: 126-34 (1996) Article DOI: 10.1021/jm950410b BindingDB Entry DOI: 10.7270/Q2VX0K7V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50368152 (CHEMBL318812) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.297 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding against muscarinic acetylcholine receptor in rat heart. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50403547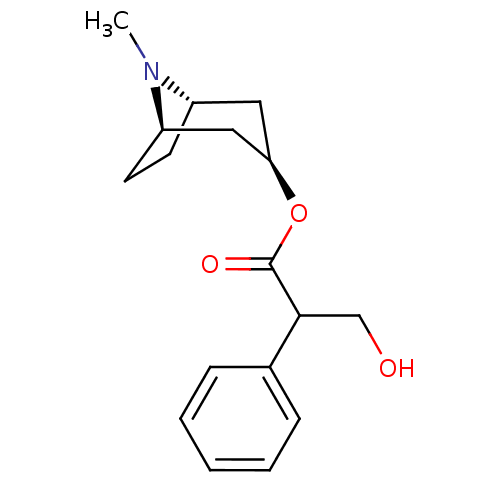 (ATROPEN | ATROPINE) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem Similars | PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description The compound was tested for inhibition of [3H]NMS binding against muscarinic acetylcholine receptor in rat brain | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1/M2/M3/M4/M5 (RAT) | BDBM50368152 (CHEMBL318812) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.302 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding against muscarinic acetylcholine receptor in rat heart. | J Med Chem 34: 3164-71 (1991) BindingDB Entry DOI: 10.7270/Q2V988Q7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM50131922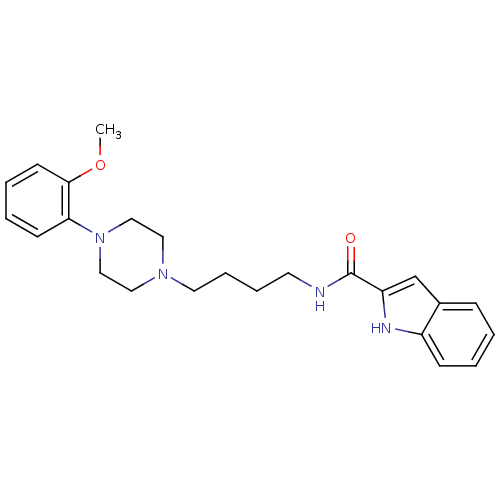 (1H-Indole-2-carboxylic acid {4-[4-(2-methoxy-pheny...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute on Drug Abuse-Intramural Research Program Curated by ChEMBL | Assay Description Displacement of [125I]IABN from human D3 receptor expressed in HEK293 cells after 60 mins by gamma counting analysis | J Med Chem 55: 6689-99 (2012) Article DOI: 10.1021/jm300482h BindingDB Entry DOI: 10.7270/Q2Q81F6B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 2C (Homo sapiens (Human)) | BDBM50086063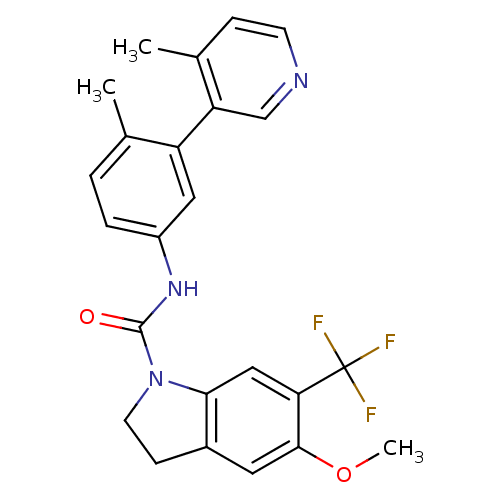 (5-Methoxy-6-trifluoromethyl-2,3-dihydro-indole-1-c...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals Curated by ChEMBL | Assay Description Binding affinity towards human cloned 5-hydroxytryptamine receptor 2C in HEK293 cells, using [3H]mesulergine as radioligand. | J Med Chem 43: 1123-34 (2000) Checked by Author BindingDB Entry DOI: 10.7270/Q23X85V5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50403547 (ATROPEN | ATROPINE) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem Similars | PubMed | 0.329 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Inhibition of [3H]QNB binding to CHO cells bearing transfected muscarinic acetylcholine receptor M3 | J Med Chem 34: 1436-40 (1991) BindingDB Entry DOI: 10.7270/Q2QC043X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 2A (Homo sapiens (Human)) | BDBM50104925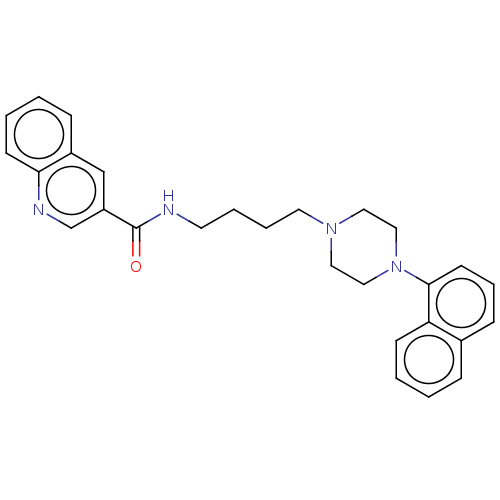 (CHEMBL3597635) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute on Drug Abuse- Intramural Research Program Curated by ChEMBL | Assay Description Displacement of [125I]DOI from 5HT2A receptor (unknown origin) | J Med Chem 58: 6195-213 (2015) Article DOI: 10.1021/acs.jmedchem.5b00776 BindingDB Entry DOI: 10.7270/Q2KS6T92 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 9228 total ) | Next | Last >> |