| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Intercellular adhesion molecule 1 |
|---|
| Ligand | BDBM50092943 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_91941 (CHEMBL704106) |
|---|
| IC50 | 240±n/a nM |
|---|
| Citation |  Liu, G; Link, JT; Pei, Z; Reilly, EB; Leitza, S; Nguyen, B; Marsh, KC; Okasinski, GF; von Geldern, TW; Ormes, M; Fowler, K; Gallatin, M Discovery of novel p-arylthio cinnamides as antagonists of leukocyte function-associated antigen-1/intracellular adhesion molecule-1 interaction. 1. Identification of an additional binding pocket based on an anilino diaryl sulfide lead. J Med Chem43:4025-40 (2000) [PubMed] Liu, G; Link, JT; Pei, Z; Reilly, EB; Leitza, S; Nguyen, B; Marsh, KC; Okasinski, GF; von Geldern, TW; Ormes, M; Fowler, K; Gallatin, M Discovery of novel p-arylthio cinnamides as antagonists of leukocyte function-associated antigen-1/intracellular adhesion molecule-1 interaction. 1. Identification of an additional binding pocket based on an anilino diaryl sulfide lead. J Med Chem43:4025-40 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Intercellular adhesion molecule 1 |
|---|
| Name: | Intercellular adhesion molecule 1 |
|---|
| Synonyms: | CD_antigen=CD54 | ICAM-1 | ICAM1 | ICAM1_HUMAN | Intercellular adhesion molecule (ICAM-1), Integrin alpha-L/beta-2 | Intercellular adhesion molecule 1 | Intercellular adhesion molecule-1 | Major group rhinovirus receptor |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 57831.10 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_826675 |
|---|
| Residue: | 532 |
|---|
| Sequence: | MAPSSPRPALPALLVLLGALFPGPGNAQTSVSPSKVILPRGGSVLVTCSTSCDQPKLLGI
ETPLPKKELLLPGNNRKVYELSNVQEDSQPMCYSNCPDGQSTAKTFLTVYWTPERVELAP
LPSWQPVGKNLTLRCQVEGGAPRANLTVVLLRGEKELKREPAVGEPAEVTTTVLVRRDHH
GANFSCRTELDLRPQGLELFENTSAPYQLQTFVLPATPPQLVSPRVLEVDTQGTVVCSLD
GLFPVSEAQVHLALGDQRLNPTVTYGNDSFSAKASVSVTAEDEGTQRLTCAVILGNQSQE
TLQTVTIYSFPAPNVILTKPEVSEGTEVTVKCEAHPRAKVTLNGVPAQPLGPRAQLLLKA
TPEDNGRSFSCSATLEVAGQLIHKNQTRELRVLYGPRLDERDCPGNWTWPENSQQTPMCQ
AWGNPLPELKCLKDGTFPLPIGESVTVTRDLEGTYLCRARSTQGEVTRKVTVNVLSPRYE
IVIITVVAAAVIMGTAGLSTYLYNRQRKIKKYRLQQAQKGTPMKPNTQATPP
|
|
|
|---|
| BDBM50092943 |
|---|
| n/a |
|---|
| Name | BDBM50092943 |
|---|
| Synonyms: | 1-(4-Acetyl-piperazin-1-yl)-3-[3-chloro-4-(2,4-dichloro-phenylsulfanyl)-phenyl]-propenone | CHEMBL101416 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H19Cl3N2O2S |
|---|
| Mol. Mass. | 469.812 |
|---|
| SMILES | CC(=O)N1CCN(CC1)C(=O)\C=C\c1ccc(Sc2ccc(Cl)cc2Cl)c(Cl)c1 |
|---|
| Structure | 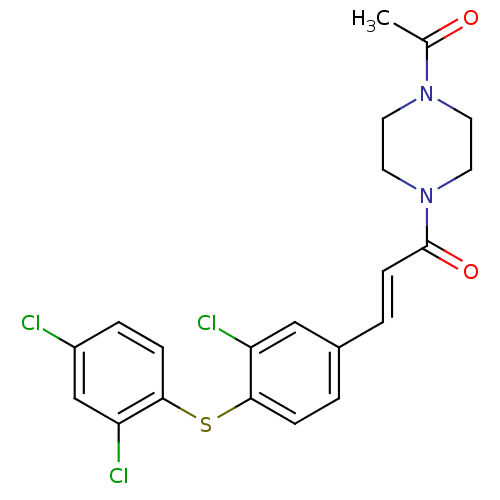
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, G; Link, JT; Pei, Z; Reilly, EB; Leitza, S; Nguyen, B; Marsh, KC; Okasinski, GF; von Geldern, TW; Ormes, M; Fowler, K; Gallatin, M Discovery of novel p-arylthio cinnamides as antagonists of leukocyte function-associated antigen-1/intracellular adhesion molecule-1 interaction. 1. Identification of an additional binding pocket based on an anilino diaryl sulfide lead. J Med Chem43:4025-40 (2000) [PubMed]
Liu, G; Link, JT; Pei, Z; Reilly, EB; Leitza, S; Nguyen, B; Marsh, KC; Okasinski, GF; von Geldern, TW; Ormes, M; Fowler, K; Gallatin, M Discovery of novel p-arylthio cinnamides as antagonists of leukocyte function-associated antigen-1/intracellular adhesion molecule-1 interaction. 1. Identification of an additional binding pocket based on an anilino diaryl sulfide lead. J Med Chem43:4025-40 (2000) [PubMed]