

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Apelin receptor | ||
| Ligand | BDBM50541318 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1984141 (CHEMBL4617547) | ||
| EC50 | 5.0±n/a nM | ||
| Citation |  Myers, MC; Bilder, DM; Cavallaro, CL; Chao, HJ; Su, S; Burford, NT; Nayeem, A; Wang, T; Yan, M; Langish, RA; Dabros, M; Li, YX; Rose, AV; Behnia, K; Onorato, JM; Gargalovic, PS; Wexler, RR; Lawrence, RM Discovery and SAR of aryl hydroxy pyrimidinones as potent small molecule agonists of the GPCR APJ. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article Myers, MC; Bilder, DM; Cavallaro, CL; Chao, HJ; Su, S; Burford, NT; Nayeem, A; Wang, T; Yan, M; Langish, RA; Dabros, M; Li, YX; Rose, AV; Behnia, K; Onorato, JM; Gargalovic, PS; Wexler, RR; Lawrence, RM Discovery and SAR of aryl hydroxy pyrimidinones as potent small molecule agonists of the GPCR APJ. Bioorg Med Chem Lett30:0 (2020) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Apelin receptor | |||
| Name: | Apelin receptor | ||
| Synonyms: | AGTRL1 | APJ | APJ_HUMAN | APLNR | Angiotensin receptor-like 1 | Apelin receptor | Apelin receptor (APJ) | G-protein coupled receptor APJ | G-protein coupled receptor HG11 | ||
| Type: | Enzyme Catalytic Domain | ||
| Mol. Mass.: | 42664.06 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P35414 | ||
| Residue: | 380 | ||
| Sequence: |
| ||
| BDBM50541318 | |||
| n/a | |||
| Name | BDBM50541318 | ||
| Synonyms: | CHEMBL4632688 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C16H20N2O3 | ||
| Mol. Mass. | 288.3416 | ||
| SMILES | CCCCc1nc(=O)cc(O)n1-c1c(C)cccc1OC |(56.29,-35.76,;57.62,-36.53,;58.95,-35.76,;60.29,-36.53,;61.62,-35.75,;61.62,-34.21,;62.96,-33.44,;62.96,-31.9,;64.29,-34.21,;64.29,-35.74,;65.63,-36.51,;62.96,-36.51,;62.96,-38.05,;64.29,-38.81,;65.62,-38.04,;64.3,-40.36,;62.96,-41.13,;61.63,-40.36,;61.63,-38.82,;60.29,-38.05,;58.96,-38.82,)| | ||
| Structure | 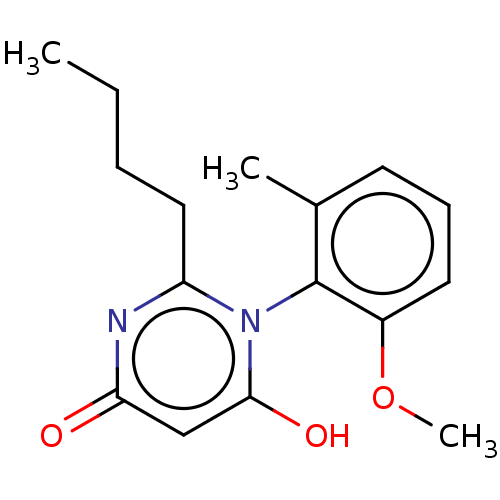
| ||