

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cytochrome P450 2D6 | ||
| Ligand | BDBM50579575 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_2135900 (CHEMBL4845510) | ||
| IC50 | >20000±n/a nM | ||
| Citation |  Nguyen, HH; Tahirovic, YA; Truax, VM; Wilson, RJ; Jecs, E; Miller, EJ; Kim, MB; Akins, NS; Xu, L; Jiang, Y; Wang, T; Sum, CS; Cvijic, ME; Schroeder, GM; Wilson, LJ; Liotta, DC Amino-Heterocycle Tetrahydroisoquinoline CXCR4 Antagonists with Improved ADME Profiles via Late-Stage Buchwald Couplings. ACS Med Chem Lett12:1605-1612 (2021) [PubMed] Article Nguyen, HH; Tahirovic, YA; Truax, VM; Wilson, RJ; Jecs, E; Miller, EJ; Kim, MB; Akins, NS; Xu, L; Jiang, Y; Wang, T; Sum, CS; Cvijic, ME; Schroeder, GM; Wilson, LJ; Liotta, DC Amino-Heterocycle Tetrahydroisoquinoline CXCR4 Antagonists with Improved ADME Profiles via Late-Stage Buchwald Couplings. ACS Med Chem Lett12:1605-1612 (2021) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cytochrome P450 2D6 | |||
| Name: | Cytochrome P450 2D6 | ||
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 | ||
| Type: | Protein | ||
| Mol. Mass.: | 55774.82 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P10635 | ||
| Residue: | 497 | ||
| Sequence: |
| ||
| BDBM50579575 | |||
| n/a | |||
| Name | BDBM50579575 | ||
| Synonyms: | CHEMBL4864332 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C26H37N5 | ||
| Mol. Mass. | 419.6055 | ||
| SMILES | CN(C[C@H]1Cc2c(CN1)cccc2N[C@H]1CC[C@H](N)CC1)[C@H]1CCCc2cccnc12 |r,wU:3.2,17.19,wD:21.23,14.15,(6.81,-26.81,;5.48,-26.03,;4.14,-26.8,;4.14,-28.34,;5.47,-29.11,;5.46,-30.65,;4.12,-31.41,;2.79,-30.64,;2.8,-29.1,;4.11,-32.95,;5.44,-33.73,;6.78,-32.96,;6.78,-31.42,;8.12,-30.66,;9.45,-31.43,;9.45,-32.97,;10.77,-33.74,;12.11,-32.98,;13.44,-33.76,;12.12,-31.44,;10.79,-30.66,;5.48,-24.49,;6.83,-23.72,;6.83,-22.17,;5.49,-21.38,;4.15,-22.16,;2.81,-21.4,;1.48,-22.17,;1.48,-23.72,;2.81,-24.49,;4.14,-23.72,)| | ||
| Structure | 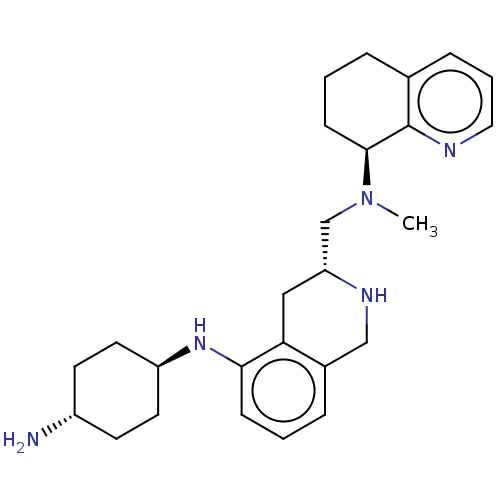
| ||