| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1/2 |
|---|
| Ligand | BDBM50022556 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_157968 (CHEMBL767498) |
|---|
| IC50 | 1800±n/a nM |
|---|
| Citation |  Clemence, F; Le Martret, O; Delevallee, F; Benzoni, J; Jouanen, A; Jouquey, S; Mouren, M; Deraedt, R 4-Hydroxy-3-quinolinecarboxamides with antiarthritic and analgesic activities. J Med Chem31:1453-62 (1988) [PubMed] Clemence, F; Le Martret, O; Delevallee, F; Benzoni, J; Jouanen, A; Jouquey, S; Mouren, M; Deraedt, R 4-Hydroxy-3-quinolinecarboxamides with antiarthritic and analgesic activities. J Med Chem31:1453-62 (1988) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1/2 |
|---|
| Name: | Prostaglandin G/H synthase 1/2 |
|---|
| Synonyms: | Cyclooxygenase |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | n/a |
|---|
| Description: | ASSAY_ID of ChEMBL is 1570295 |
|---|
| Components: | This complex has 2 components. |
|---|
| Component 1 |
| Name: | Prostaglandin G/H synthase 2 |
|---|
| Synonyms: | COX2 | Cyclooxygenase | Cyclooxygenase-2 | PGH synthase 2 | PGH2_BOVIN | PGHS-2 | PHS II | PTGS2 | Prostaglandin H2 synthase 2 | Prostaglandin-endoperoxide synthase 2 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 69173.20 |
|---|
| Organism: | Bos taurus |
|---|
| Description: | ChEMBL_157990 |
|---|
| Residue: | 604 |
|---|
| Sequence: | MLARALLLCAAVALSGAANPCCSHPCQNRGVCMSVGFDQYKCDCTRTGFYGENCTTPEFL
TRIKLLLKPTPNTVHYILTHFKGVWNIVNKISFLRNMIMRYVLTSRSHLIESPPTYNVHY
SYKSWEAFSNLSYYTRALPPVPDDCPTPMGVKGRKELPDSKEVVKKVLLRRKFIPDPQGT
NLMFAFFAQHFTHQFFKTDFERGPAFTKGKNHGVDLSHIYGESLERQHKLRLFKDGKMKY
QMINGEMYPPTVKDTQVEMIYPPHVPEHLKFAVGQEVFGLVPGLMMYATIWLREHNRVCD
VLKQEHPEWGDEQLFQTSRLILIGETIKIVIEDYVQHLSGYHFKLKFDPELLFNQQFQYQ
NRIAAEFNTLYHWHPLLPDVFQIDGQEYNYQQFIYNNSVLLEHGLTQFVESFTRQRAGRV
AGGRNLPVAVEKVSKASIDQSREMKYQSFNEYRKRFLVKPYESFEELTGEKEMAAELEAL
YGDIDAMEFYPALLVEKPRPDAIFGETMVEAGAPFSLKGLMGNPICSPEYWKPSTFGGEV
GFKIINTASIQSLICSNVKGCPFTSFSVQDTHLTKTVTINASSSHSGLDDINPTVLLKER
STEL
|
|
|
|---|
| Component 2 |
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | COX-1 | COX1 | Cyclooxygenase | Cyclooxygenase-1 | PGH1_BOVIN | PTGS1 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 68814.10 |
|---|
| Organism: | Bos taurus |
|---|
| Description: | ChEMBL_547440 |
|---|
| Residue: | 600 |
|---|
| Sequence: | MSRQGISLRFPLLLLLLSPSPVLPADPGAPAPVNPCCYYPCQHQGICVRFGLDRYQCDCT
RTGYYGPNCTIPEIWTWLRTTLRPSPSFVHFLLTHGRWLWDFVNATFIRDKLMRLVLTVR
SNLIPSPPTYNVAHDYISWESFSNVSYYTRILPSVPRDCPTPMGTKGKKQLPDAEFLSRR
FLLRRKFIPDPQGTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLER
QYQLRLFKDGKLKYQMLNGEVYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLMV
YATIWLREHNRVCDLLKAEHPTWGDEQLFQTARLILIGETIKIVIEEYVQQLSGYFLQLK
FDPELLFGAQFQYRNRIAMEFNQLYHWHPLMPDSFRVGPQDYSYEQFLFNTSMLVDYGVE
ALVDAFSRQPAGRIGGGRNIDHHILHVAVDVIKESRELRLQPFNEYRKRFGMKPYTSFQE
LTGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEMGAPFSLKGLLGNPIC
SPEYWKASTFGGDVGFNLVKTATLKKLVCLNTKTCPYVSFHVPDPHREDRPGVERPPTEL
|
|
|
|---|
| BDBM50022556 |
|---|
| n/a |
|---|
| Name | BDBM50022556 |
|---|
| Synonyms: | 2-Dichloromethyl-4-hydroxy-8-trifluoromethyl-quinoline-3-carboxylic acid thiazol-2-ylamide | CHEMBL285278 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H8Cl2F3N3O2S |
|---|
| Mol. Mass. | 422.209 |
|---|
| SMILES | Oc1c(C(=O)Nc2nccs2)c(nc2c(cccc12)C(F)(F)F)C(Cl)Cl |
|---|
| Structure | 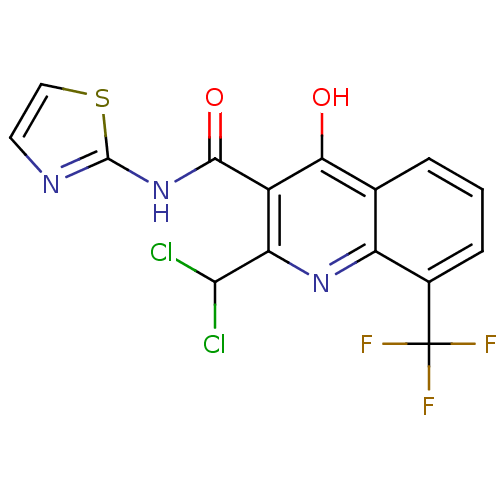
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Clemence, F; Le Martret, O; Delevallee, F; Benzoni, J; Jouanen, A; Jouquey, S; Mouren, M; Deraedt, R 4-Hydroxy-3-quinolinecarboxamides with antiarthritic and analgesic activities. J Med Chem31:1453-62 (1988) [PubMed]
Clemence, F; Le Martret, O; Delevallee, F; Benzoni, J; Jouanen, A; Jouquey, S; Mouren, M; Deraedt, R 4-Hydroxy-3-quinolinecarboxamides with antiarthritic and analgesic activities. J Med Chem31:1453-62 (1988) [PubMed]