| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50084346 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_158914 (CHEMBL763302) |
|---|
| IC50 | 42400±n/a nM |
|---|
| Citation |  Puig, C; Crespo, MI; Godessart, N; Feixas, J; Ibarzo, J; Jiménez, JM; Soca, L; Cardelús, I; Heredia, A; Miralpeix, M; Puig, J; Beleta, J; Huerta, JM; López, M; Segarra, V; Ryder, H; Palacios, JM Synthesis and biological evaluation of 3,4-diaryloxazolones: A new class of orally active cyclooxygenase-2 inhibitors. J Med Chem43:214-23 (2000) [PubMed] Puig, C; Crespo, MI; Godessart, N; Feixas, J; Ibarzo, J; Jiménez, JM; Soca, L; Cardelús, I; Heredia, A; Miralpeix, M; Puig, J; Beleta, J; Huerta, JM; López, M; Segarra, V; Ryder, H; Palacios, JM Synthesis and biological evaluation of 3,4-diaryloxazolones: A new class of orally active cyclooxygenase-2 inhibitors. J Med Chem43:214-23 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | COX-1 | COX1 | Cyclooxygenase | Cyclooxygenase-1 | Cyclooxygenase-1 (COX-1) | PGH1_HUMAN | PTGS1 | Prostaglandin G/H synthase (cyclooxygenase) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68692.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P23219 |
|---|
| Residue: | 599 |
|---|
| Sequence: | MSRSLLLWFLLFLLLLPPLPVLLADPGAPTPVNPCCYYPCQHQGICVRFGLDRYQCDCTR
TGYSGPNCTIPGLWTWLRNSLRPSPSFTHFLLTHGRWFWEFVNATFIREMLMRLVLTVRS
NLIPSPPTYNSAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDAQLLARRF
LLRRKFIPDPQGTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLERQ
YQLRLFKDGKLKYQVLDGEMYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLMLY
ATLWLREHNRVCDLLKAEHPTWGDEQLFQTTRLILIGETIKIVIEEYVQQLSGYFLQLKF
DPELLFGVQFQYRNRIAMEFNHLYHWHPLMPDSFKVGSQEYSYEQFLFNTSMLVDYGVEA
LVDAFSRQIAGRIGGGRNMDHHILHVAVDVIRESREMRLQPFNEYRKRFGMKPYTSFQEL
VGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEIGAPFSLKGLLGNPICS
PEYWKPSTFGGEVGFNIVKTATLKKLVCLNTKTCPYVSFRVPDASQDDGPAVERPSTEL
|
|
|
|---|
| BDBM50084346 |
|---|
| n/a |
|---|
| Name | BDBM50084346 |
|---|
| Synonyms: | 3-(4-Ethyl-phenyl)-4-(4-methanesulfonyl-phenyl)-3H-oxazol-2-one | CHEMBL306716 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H17NO4S |
|---|
| Mol. Mass. | 343.397 |
|---|
| SMILES | CCc1ccc(cc1)-n1c(coc1=O)-c1ccc(cc1)S(C)(=O)=O |
|---|
| Structure | 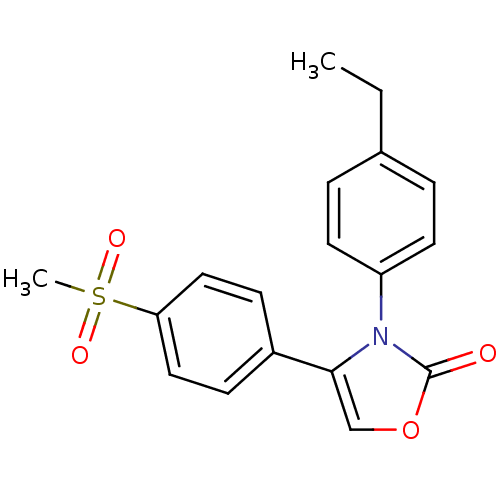
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Puig, C; Crespo, MI; Godessart, N; Feixas, J; Ibarzo, J; Jiménez, JM; Soca, L; Cardelús, I; Heredia, A; Miralpeix, M; Puig, J; Beleta, J; Huerta, JM; López, M; Segarra, V; Ryder, H; Palacios, JM Synthesis and biological evaluation of 3,4-diaryloxazolones: A new class of orally active cyclooxygenase-2 inhibitors. J Med Chem43:214-23 (2000) [PubMed]
Puig, C; Crespo, MI; Godessart, N; Feixas, J; Ibarzo, J; Jiménez, JM; Soca, L; Cardelús, I; Heredia, A; Miralpeix, M; Puig, J; Beleta, J; Huerta, JM; López, M; Segarra, V; Ryder, H; Palacios, JM Synthesis and biological evaluation of 3,4-diaryloxazolones: A new class of orally active cyclooxygenase-2 inhibitors. J Med Chem43:214-23 (2000) [PubMed]