

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Vasopressin V1a receptor | ||
| Ligand | BDBM316350 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | Binding Assay | ||
| Ki | 5.5±n/a nM | ||
| Citation |  Braje, W; Froggett, J; Geneste, H; Hornberger, W; Jantos, K; Kling, A; van Gaalen, M Fused heterocyclic or carbocyclic compounds carrying a substituted cycloaliphatic radical and use thereof for treating vasopressin-related diseases US Patent US9617226 Publication Date 4/11/2017 Braje, W; Froggett, J; Geneste, H; Hornberger, W; Jantos, K; Kling, A; van Gaalen, M Fused heterocyclic or carbocyclic compounds carrying a substituted cycloaliphatic radical and use thereof for treating vasopressin-related diseases US Patent US9617226 Publication Date 4/11/2017 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Vasopressin V1a receptor | |||
| Name: | Vasopressin V1a receptor | ||
| Synonyms: | AVPR V1a | AVPR1 | AVPR1A | Antidiuretic hormone receptor 1a | V1AR_HUMAN | V1aR | VASOPRESSIN V1A | Vascular/hepatic-type arginine vasopressin receptor | Vasopressin V1 receptor | Vasopressin V1a receptor | Vasopressin receptor | ||
| Type: | Receptor | ||
| Mol. Mass.: | 46820.18 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P37288 | ||
| Residue: | 418 | ||
| Sequence: |
| ||
| BDBM316350 | |||
| n/a | |||
| Name | BDBM316350 | ||
| Synonyms: | US9617226, Example 31 | cis-2-[2-(3-Chlorophenyl)-6-[3-(6-oxa-2-azaspiro[3.3]heptan-2-ylmethyl)cyclobutyl]-4-oxo-quinazolin-3-yl]-N-isopropyl-acetamide | ||
| Type | Small organic molecule | ||
| Emp. Form. | C29H33ClN4O3 | ||
| Mol. Mass. | 521.05 | ||
| SMILES | CC(C)NC(=O)Cn1c(nc2ccc(cc2c1=O)[C@@H]1C[C@H](CN2CC3(COC3)C2)C1)-c1cccc(Cl)c1 |r,wD:18.19,20.22,(3.74,-2.34,;2.97,-3.67,;3.74,-5.01,;1.43,-3.67,;.66,-2.34,;1.43,-1.01,;-.88,-2.34,;-1.65,-1.01,;-3.19,-1.01,;-3.96,.33,;-3.19,1.66,;-3.96,3,;-3.19,4.33,;-1.65,4.33,;-.88,3,;-1.65,1.66,;-.88,.33,;.66,.33,;-.88,5.66,;-1.28,7.15,;.21,7.55,;.98,8.88,;2.52,8.88,;3.61,9.97,;4.7,8.88,;5.79,9.97,;6.88,8.88,;5.79,7.79,;3.61,7.79,;.61,6.06,;-3.96,-2.34,;-3.19,-3.67,;-3.96,-5.01,;-5.5,-5.01,;-6.27,-3.67,;-7.81,-3.67,;-5.5,-2.34,)| | ||
| Structure | 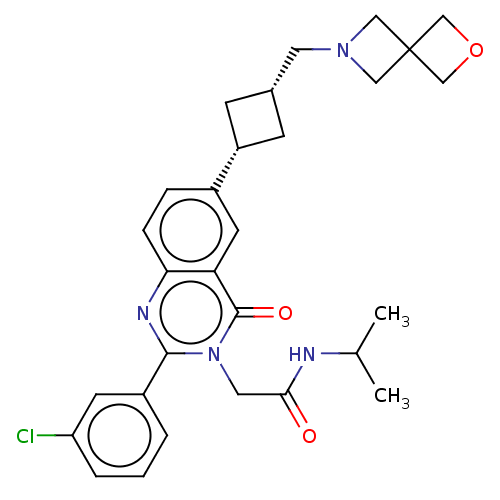
| ||