| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM12398 |
|---|
| Substrate/Competitor | BDBM12500 |
|---|
| Meas. Tech. | Factor Xa Inhibition Assay |
|---|
| Ki | 89±n/a nM |
|---|
| Citation |  Nazare, M; Will, DW; Matter, H; Schreuder, H; Ritter, K; Urmann, M; Essrich, M; Bauer, A; Wagner, M; Czech, J; Lorenz, M; Laux, V; Wehner, V Probing the subpockets of factor Xa reveals two binding modes for inhibitors based on a 2-carboxyindole scaffold: a study combining structure-activity relationship and X-ray crystallography. J Med Chem48:4511-25 (2005) [PubMed] Article Nazare, M; Will, DW; Matter, H; Schreuder, H; Ritter, K; Urmann, M; Essrich, M; Bauer, A; Wagner, M; Czech, J; Lorenz, M; Laux, V; Wehner, V Probing the subpockets of factor Xa reveals two binding modes for inhibitors based on a 2-carboxyindole scaffold: a study combining structure-activity relationship and X-ray crystallography. J Med Chem48:4511-25 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM12398 |
|---|
| BDBM12500 |
|---|
| Name | BDBM12398 |
|---|
| Synonyms: | 1-(3-Methoxy-benzyl)-1H-indole-2-carboxylic acid (1-isopropyl-piperidin-4-yl)-amide | 1-[(3-methoxyphenyl)methyl]-N-[1-(propan-2-yl)piperidin-4-yl]-1H-indole-2-carboxamide | 2-Carboxyindole Scaffold 43 | 2-carboxyindole 12 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H31N3O2 |
|---|
| Mol. Mass. | 405.5325 |
|---|
| SMILES | COc1cccc(Cn2c(cc3ccccc23)C(=O)NC2CCN(CC2)C(C)C)c1 |
|---|
| Structure | 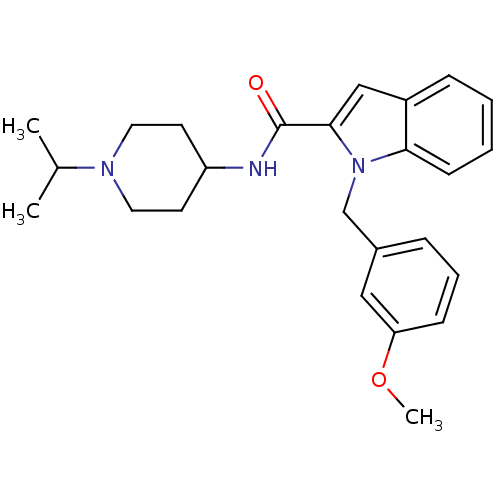
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nazare, M; Will, DW; Matter, H; Schreuder, H; Ritter, K; Urmann, M; Essrich, M; Bauer, A; Wagner, M; Czech, J; Lorenz, M; Laux, V; Wehner, V Probing the subpockets of factor Xa reveals two binding modes for inhibitors based on a 2-carboxyindole scaffold: a study combining structure-activity relationship and X-ray crystallography. J Med Chem48:4511-25 (2005) [PubMed] Article
Nazare, M; Will, DW; Matter, H; Schreuder, H; Ritter, K; Urmann, M; Essrich, M; Bauer, A; Wagner, M; Czech, J; Lorenz, M; Laux, V; Wehner, V Probing the subpockets of factor Xa reveals two binding modes for inhibitors based on a 2-carboxyindole scaffold: a study combining structure-activity relationship and X-ray crystallography. J Med Chem48:4511-25 (2005) [PubMed] Article